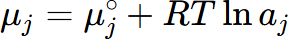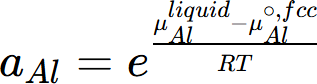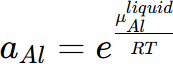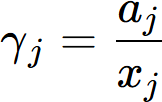Activity and Activity Coefficient
The activity of a component j, aj, is defined by
where μj is the chemical potential of the component j in equilibrium state and 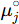 is the chemical potential of this component at its reference state. For example, the activity of component Al in the liquid Al-Mg system referring the Al in Fcc phase is calculated by
is the chemical potential of this component at its reference state. For example, the activity of component Al in the liquid Al-Mg system referring the Al in Fcc phase is calculated by
The corresponding Table column name is a(Al@Liquid:Fcc[Al]), or a(Al@Liquid:Fcc).
If a reference state is not specified in Table column name, the default reference state in the database is taken as the reference state. For example, a(Al@Liquid) is calculated by
Activity coefficient is defined as
which is available from Table by defining Table column name similar to that of activity. Table column name for the activity coefficient of Al in liquid is r(Al@Liquid:Fcc[Al]), or r(Al@Liquid:Fcc).
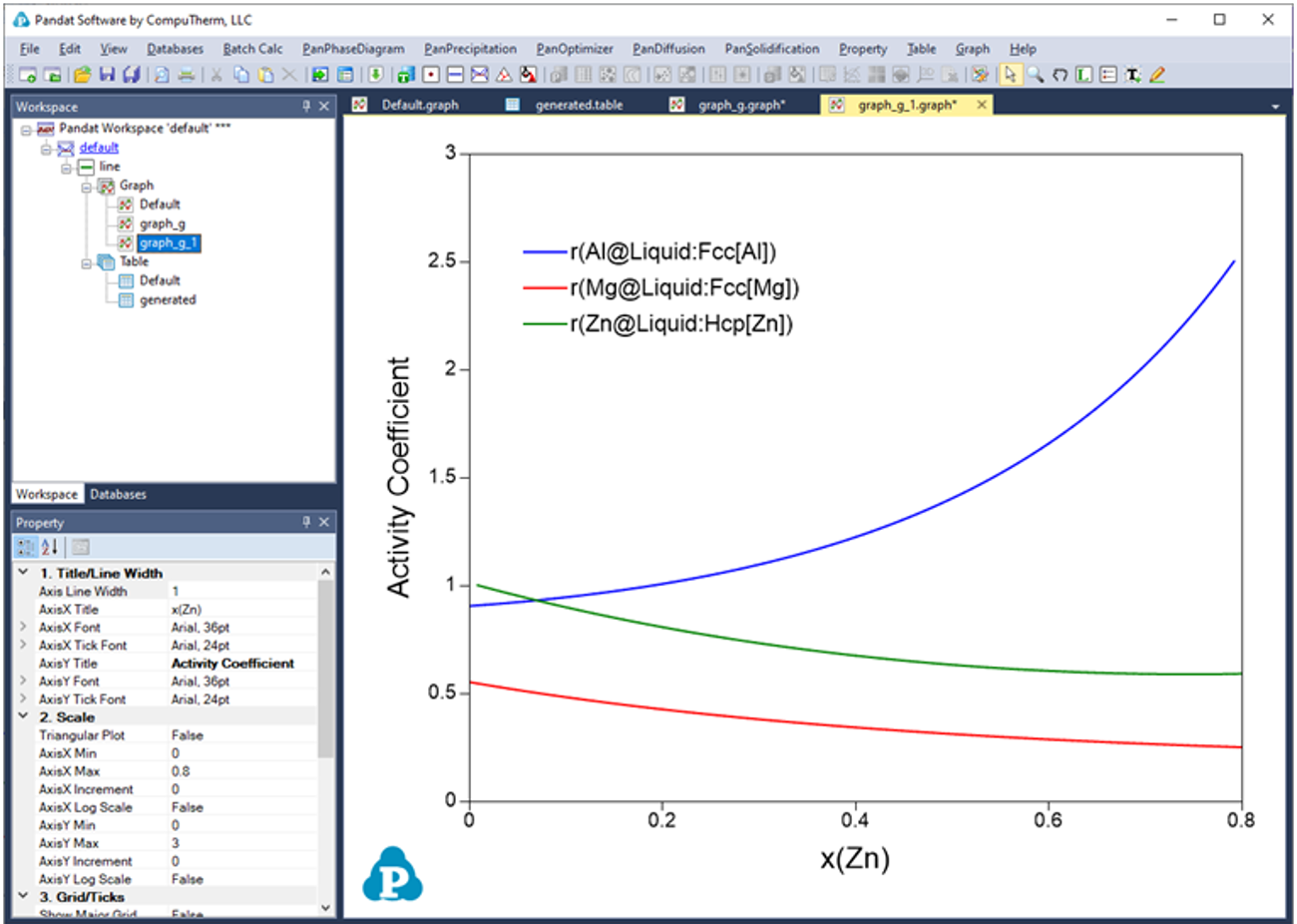
Below shows how to creating a table of activity and activity coefficient from a line calculation result at 1000K in the Al-Mg-Zn ternary system. The two end points are at x(Mg)=0.2,x(Al)=0.8 and x(Mg)=0.2,x(Zn)=0.8. The liquid phase is the stable phase under this condition. The setting in Figure 3.68 is to choose Fcc Al, Fcc Mg, and Hcp Zn as the reference state for the activities and activity coefficients. Figure 3.69 shows the created table. Figure 3.70 and Figure 3.71 shows the plotted activity and activity coefficient vs x(Zn).
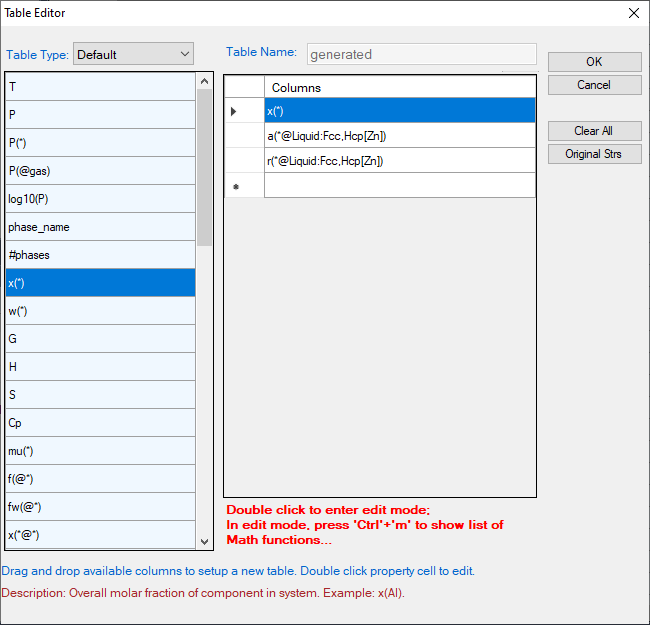
Figure 3.68: Table editor for creating activity and activity coefficient table
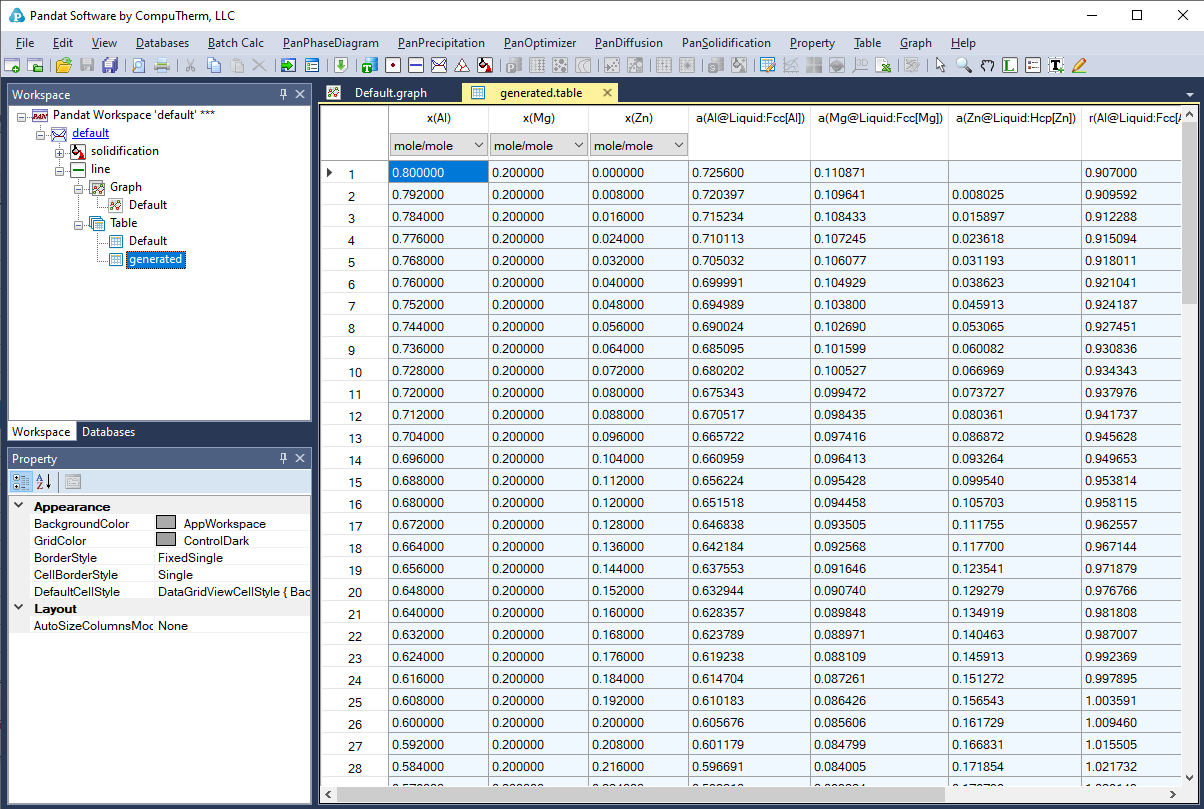
Figure 3.69: Table of activity and activity coefficient
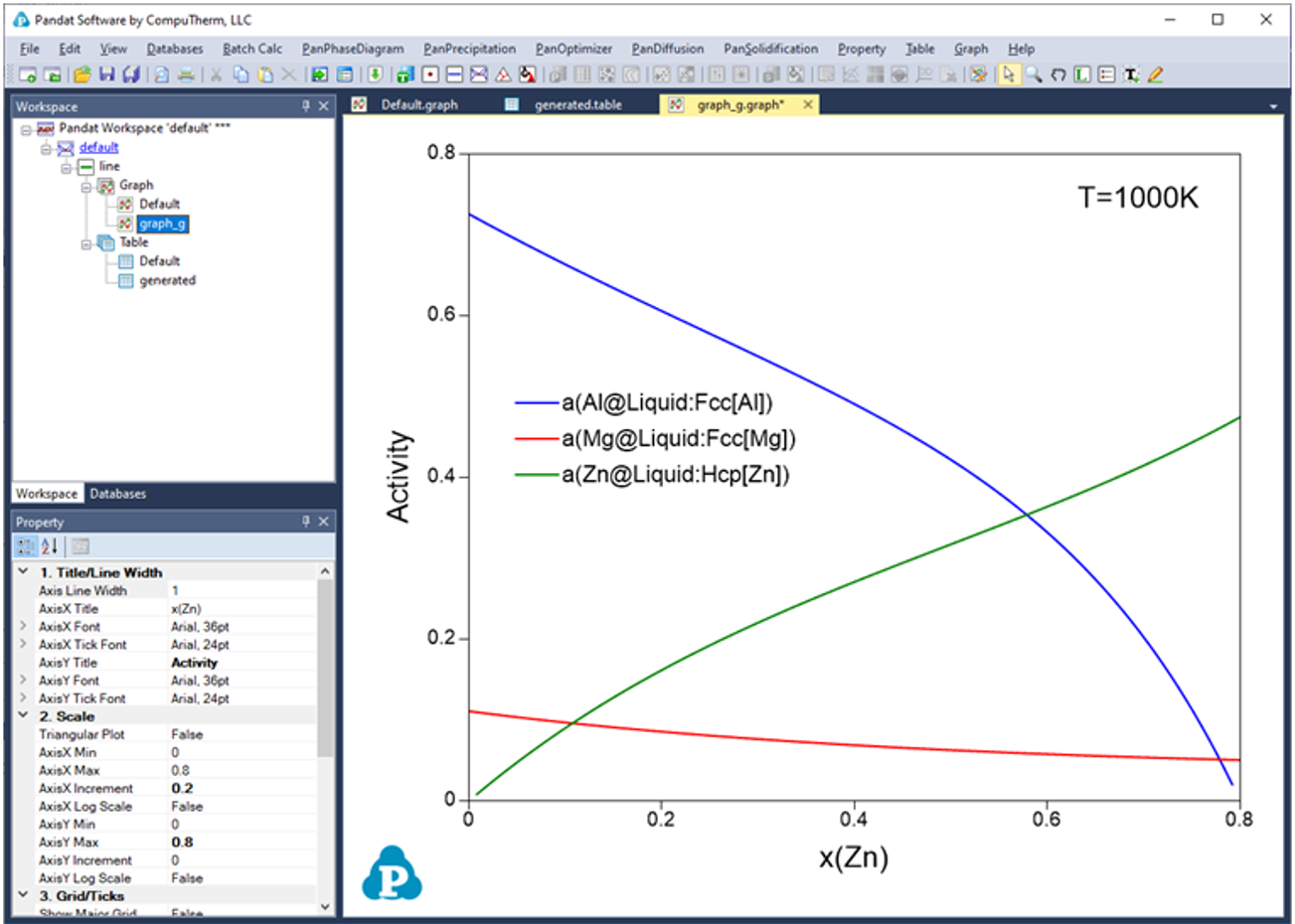
Figure 3.70: Graph of activity vs. x(Zn)