Thermodynamic Database
Components (40)
Total of 40 components are included in the database as listed here:
Major alloying elements: Al, Cu, Fe, Mg, Mn, Si and Zn
Minor alloying elements: Ag, B, Ba, Be, Bi, C, Ca, Ce, Co, Cr, Er, Gd, Ge, Hf, In, K, La, Li, Mo, Na, Nb, Ni, O, Pb, Sb, Sc, Sn, Sr, Ti, V, W, Y, and Zr
Suggested Composition Range
The suggested composition range for each element is listed in Table 1.1. It should be noted that this given composition range is rather conservative. It is derived from the chemistries of the multicomponent commercial alloys that have been used to validate the current database. In the subsystems, many of these elements can be applied to a much wider composition range. In fact, some binary and ternary systems have been fully assessed and they are covered in the full range from 0-100% as given in Section Assessed Subsystems.
Table 1.1: Suggested composition range
|
Elements |
Composition Range (wt.%) |
|
Al |
80 ~ 100 |
|
Cu |
0 ~ 5.5 |
|
Fe |
0 ~ 2.0 |
|
Mg |
0 ~ 7.6 |
|
Mn |
0 ~ 1.2 |
|
Si |
0 ~ 17.5 |
|
Zn |
0 ~ 8.1 |
|
Others |
0 ~ 1 |
What is new in PanAl2026
- Addition of the component Mo
- Introducing solubility of Fe with the Al-Cr-Si intermetallic phases
- Improve the diffusion of rare earth elements within (Al)
- Improve the Pb-related systems and the Al-Mn-Sc ternary system
Phases
Total of 1236 phases are included in the current database. The names and thermodynamic models of some phases are given in Table 1.2. Information on all the other phases is listed in PanAl2026: List of Phases. Users can also view it through TDB viewer of Pandat.
Table 1.2: Phase name and related information
|
Name |
Lattice Size |
Constituent |
|
Ag5Zn8 |
(2)(2)(3)(6) |
(Ag,Zn)(Ag)(Ag,Zn)(Ag,Zn) |
|
AgMg3 |
(0.23)(0.77) |
(Ag,Cu)(Al,Mg) |
|
AgMg4 |
(0.2)(0.8) |
(Ag,Cu)(Al,Mg) |
|
Al18Mg3V2 |
(18)(3)(2) |
(Al)(Mg)(V) |
|
Al2Fe |
(2)(1) |
(Al)(Cr,Fe,Mn) |
|
Al2LiMg |
(0.53)(0.33)(0.14) |
(Al)(Li)(Mg) |
|
Al2W |
(2)(1) |
(Al)(W) |
|
Al5Fe2 |
(5)(2) |
(Al)(Cr,Fe,Mn) |
|
Al5Fe4 |
(1) |
(Al,Fe,Mn) |
|
Al8FeMg3Si6 |
(8)(3)(1)(6) |
(Al)(Mg)(Fe)(Si) |
|
Al8FeMnSi2 |
(16)(2)(2)(3) |
(Al)(Fe)(Mn)(Si) |
|
AlCu_Theta |
(0.667)(0.333) |
(Al)(Ag,Al,Cu) |
|
AlMg_Gamma |
(10)(24)(24) |
(Ca,Mg)(Al,Cu,Li,Mg,Zn)(Al,Cu,Mg,Zn) |
|
Alpha_AlFeSi |
(0.66)(0.19)(0.05)(0.1) |
(Al)(Fe)(Si)(Al,Si) |
|
Cu3Mg2Si |
(3)(2)(1) |
(Cu)(Mg)(Si) |
|
Delta_AlFeSi |
(5)(1) |
(Al,Si)(Fe) |
|
Fcc |
(1)(1) |
(Ag,Al,Ba,Be,Bi,Ca,Ce,Co,Cr,Cu,Er,Fe, |
|
Gamma_AlFeSi |
(0.635)(0.205)(0.16) |
(Al)(Fe)(Si) |
|
Li2MgSi |
(0.5)(0.25)(0.25) |
(Li)(Mg)(Si) |
|
LiMg2Si |
(0.5)(0.25)(0.25) |
(Mg)(Si)(Li,Va) |
|
Mu |
(7)(2)(4) |
(Co,Fe,Mn,Nb,W)(Nb,W)(Co,Fe,Nb,W) |
|
NbSn2 |
(1)(2) |
(Nb)(Sn) |
|
Q_Al5Cu2Mg8Si6 |
(0.2381)(0.0952)(0.381)(0.2857) |
(Al)(Cu)(Mg)(Si) |
|
T10_AlFeSi |
(0.6)(0.25)(0.15) |
(Al)(Fe)(Si) |
Assessed Subsystems
A total of 581 subsystems, including 492 binary and 89 ternary subsystems have been assessed. The modeling status is indicated by numbers. The systems with number 10 are fully assessed in the whole composition range. The higher value shows higher reliability of the system.
|
Ag-Al(10) |
Ag-B(10) |
Ag-Bi(10) |
Ag-C(10) |
Ag-Ca(10) |
Ag-Ce(10) |
Ag-Co(10) |
|
Ag-Cr(10) |
Ag-Cu(10) |
Ag-Er(10) |
Ag-Fe(10) |
Ag-Gd(10) |
Ag-Ge(10) |
Ag-In(10) |
|
Ag-La(10) |
Ag-Mg(10) |
Ag-Mo(10) |
Ag-Na(10) |
Ag-Ni(10) |
Ag-O(5) |
Ag-Pb(10) |
|
Ag-Sb(10) |
Ag-Sc(10) |
Ag-Si(10) |
Ag-Sn(10) |
Ag-Sr(10) |
Ag-Ti(10) |
Ag-V(10) |
|
Ag-W(10) |
Ag-Y(10) |
Ag-Zn(10) |
Ag-Zr(10) |
Al-B(10) |
Al-Ba(10) |
Al-Be(10) |
|
Al-Bi(10) |
Al-C(10) |
Al-Ca(10) |
Al-Ce(10) |
Al-Co(10) |
Al-Cr(10) |
Al-Cu(10) |
|
Al-Er(10) |
Al-Fe(10) |
Al-Gd(10) |
Al-Ge(10) |
Al-Hf(10) |
Al-In(10) |
Al-K(10) |
|
Al-La(10) |
Al-Li(10) |
Al-Mg(10) |
Al-Mn(10) |
Al-Mo(10) |
Al-Na(10) |
Al-Nb(10) |
|
Al-Ni(10) |
Al-O(10) |
Al-Pb(10) |
Al-Sb(10) |
Al-Sc(10) |
Al-Si(10) |
Al-Sn(10) |
|
Al-Sr(10) |
Al-Ti(10) |
Al-V(10) |
Al-W(10) |
Al-Y(10) |
Al-Zn(10) |
Al-Zr(10) |
|
B-C(10) |
B-Ca(10) |
B-Co(10) |
B-Cr(10) |
B-Cu(10) |
B-Er(10) |
B-Fe(10) |
|
B-Hf(10) |
B-La(10) |
B-Mg(10) |
B-Mn(10) |
B-Mo(10) |
B-Nb(10) |
B-Ni(10) |
|
B-O(10) |
B-Sc(10) |
B-Si(10) |
B-Sn(10) |
B-Sr(10) |
B-Ti(10) |
B-V(10) |
|
B-W(10) |
B-Zr(10) |
Ba-Be(10) |
Ba-Bi(10) |
Ba-Ca(10) |
Ba-Cr(10) |
Ba-Cu(10) |
|
Ba-Fe(10) |
Ba-Gd(10) |
Ba-Ge(10) |
Ba-La(10) |
Ba-Li(10) |
Ba-Mg(10) |
Ba-Mn(10) |
|
Ba-Ni(10) |
Ba-O(10) |
Ba-Pb(10) |
Ba-Sc(10) |
Ba-Si(10) |
Ba-Sr(10) |
Ba-Ti(10) |
|
Ba-V(10) |
Ba-Y(10) |
Ba-Zn(10) |
Be-O(10) |
Be-Si(10) |
Bi-Cr(10) |
Bi-Cu(10) |
|
Bi-Er(10) |
Bi-Fe(10) |
Bi-Gd(10) |
Bi-Ge(10) |
Bi-In(10) |
Bi-La(10) |
Bi-Mg(10) |
|
Bi-Mn(10) |
Bi-Ni(10) |
Bi-O(10) |
Bi-Pb(10) |
Bi-Sb(10) |
Bi-Si(10) |
Bi-Sn(10) |
|
Bi-Ti(10) |
Bi-Zn(10) |
C-Ce(10) |
C-Co(10) |
C-Cr(10) |
C-Fe(10) |
C-Ge(10) |
|
C-Li(10) |
C-Mg(10) |
C-Mn(10) |
C-Mo(10) |
C-Nb(10) |
C-Ni(10) |
C-O(10) |
|
C-Si(10) |
C-V(10) |
C-W(10) |
C-Zr(10) |
Ca-Ce(10) |
Ca-Cu(10) |
Ca-Er(10) |
|
Ca-Fe(10) |
Ca-Gd(10) |
Ca-La(10) |
Ca-Li(10) |
Ca-Mg(10) |
Ca-Mn(10) |
Ca-Na(10) |
|
Ca-Ni(10) |
Ca-O(10) |
Ca-Pb(10) |
Ca-Sc(10) |
Ca-Si(10) |
Ca-Sn(10) |
Ca-Sr(10) |
|
Ca-Zn(10) |
Ce-Co(10) |
Ce-Cu(10) |
Ce-Er(10) |
Ce-Fe(10) |
Ce-La(10) |
Ce-Li(10) |
|
Ce-Mg(10) |
Ce-Mn(10) |
Ce-Mo(10) |
Ce-Ni(10) |
Ce-O(10) |
Ce-Sb(10) |
Ce-Sc(10) |
|
Ce-Si(10) |
Ce-Ti(10) |
Ce-V(10) |
Ce-Y(10) |
Ce-Zn(10) |
Co-Cr(10) |
Co-Cu(10) |
|
Co-Er(10) |
Co-Fe(10) |
Co-Ge(10) |
Co-In(10) |
Co-La(10) |
Co-Mn(10) |
Co-Mo(10) |
|
Co-Nb(10) |
Co-Ni(10) |
Co-O(10) |
Co-Sb(10) |
Co-Sc(10) |
Co-Si(10) |
Co-Sn(10) |
|
Co-W(10) |
Co-Y(10) |
Co-Zn(10) |
Cr-Cu(10) |
Cr-Fe(10) |
Cr-Ge(10) |
Cr-Hf(10) |
|
Cr-La(10) |
Cr-Mg(10) |
Cr-Mn(10) |
Cr-Mo(10) |
Cr-Na(10) |
Cr-Nb(10) |
Cr-Ni(10) |
|
Cr-O(10) |
Cr-Sc(10) |
Cr-Si(10) |
Cr-Sn(10) |
Cr-Ti(10) |
Cr-V(10) |
Cr-W(10) |
|
Cr-Y(10) |
Cr-Zn(10) |
Cr-Zr(10) |
Cu-Er(10) |
Cu-Fe(10) |
Cu-Gd(10) |
Cu-Ge(10) |
|
Cu-Hf(10) |
Cu-In(10) |
Cu-La(10) |
Cu-Li(10) |
Cu-Mg(10) |
Cu-Mn(10) |
Cu-Mo(10) |
|
Cu-Na(10) |
Cu-Nb(10) |
Cu-Ni(10) |
Cu-O(10) |
Cu-Pb(10) |
Cu-Sb(10) |
Cu-Sc(10) |
|
Cu-Si(10) |
Cu-Sn(10) |
Cu-Sr(10) |
Cu-Ti(10) |
Cu-V(10) |
Cu-W(10) |
Cu-Y(10) |
|
Cu-Zn(10) |
Cu-Zr(10) |
Er-Fe(10) |
Er-Gd(10) |
Er-Ge(10) |
Er-Hf(10) |
Er-La(10) |
|
Er-Li(10) |
Er-Mg(10) |
Er-Mn(10) |
Er-Nb(10) |
Er-O(5) |
Er-Sc(10) |
Er-Si(10) |
|
Er-Ti(10) |
Er-V(10) |
Er-W(10) |
Er-Y(10) |
Er-Zn(10) |
Er-Zr(10) |
Fe-Gd(10) |
|
Fe-Hf(10) |
Fe-In(10) |
Fe-La(10) |
Fe-Mg(10) |
Fe-Mn(10) |
Fe-Mo(10) |
Fe-Nb(10) |
|
Fe-Ni(10) |
Fe-O(10) |
Fe-Pb(10) |
Fe-Sb(10) |
Fe-Sc(10) |
Fe-Si(10) |
Fe-Sn(10) |
|
Fe-Sr(10) |
Fe-Ti(10) |
Fe-V(10) |
Fe-W(10) |
Fe-Y(10) |
Fe-Zn(10) |
Fe-Zr(10) |
|
Gd-Mg(10) |
Gd-Mn(10) |
Gd-Ni(10) |
Gd-O(5) |
Gd-Sb(10) |
Gd-Sc(10) |
Gd-Si(10) |
|
Gd-Ti(10) |
Gd-Zn(10) |
Gd-Zr(10) |
Ge-Hf(10) |
Ge-In(10) |
Ge-K(10) |
Ge-Mg(10) |
|
Ge-Mn(10) |
Ge-Mo(10) |
Ge-Na(10) |
Ge-Nb(10) |
Ge-Ni(10) |
Ge-O(10) |
Ge-Pb(10) |
|
Ge-Sb(10) |
Ge-Sc(10) |
Ge-Si(10) |
Ge-Sn(10) |
Ge-Sr(10) |
Ge-Ti(10) |
Ge-V(10) |
|
Ge-W(8) |
Ge-Zn(10) |
Hf-Mn(10) |
Hf-Mo(10) |
Hf-Nb(10) |
Hf-Ni(10) |
Hf-O(10) |
|
Hf-Si(10) |
Hf-Sn(10) |
Hf-Ti(10) |
Hf-W(10) |
Hf-Zr(10) |
In-Ni(10) |
In-Pb(10) |
|
In-Sb(10) |
In-Si(10) |
In-Sn(10) |
In-V(10) |
In-Zn(10) |
K-Na(10) |
K-O(5) |
|
La-Mg(10) |
La-Mn(10) |
La-Mo(10) |
La-Nb(10) |
La-Ni(10) |
La-O(5) |
La-Pb(10) |
|
La-Sb(10) |
La-Sc(10) |
La-Si(10) |
La-Sn(10) |
La-Ti(10) |
La-V(10) |
La-W(10) |
|
La-Zn(10) |
La-Zr(10) |
Li-Mg(10) |
Li-Mn(10) |
Li-Na(10) |
Li-O(10) |
Li-Sc(10) |
|
Li-Si(10) |
Li-Sn(10) |
Li-Sr(10) |
Li-Zn(10) |
Li-Zr(10) |
Mg-Mn(10) |
Mg-Na(10) |
|
Mg-Ni(10) |
Mg-O(10) |
Mg-Sb(10) |
Mg-Sc(10) |
Mg-Si(10) |
Mg-Sn(10) |
Mg-Sr(10) |
|
Mg-Ti(10) |
Mg-Y(10) |
Mg-Zn(10) |
Mg-Zr(10) |
Mn-Mo(10) |
Mn-Nb(10) |
Mn-Ni(10) |
|
Mn-O(10) |
Mn-Pb(10) |
Mn-Sc(10) |
Mn-Si(10) |
Mn-Sn(10) |
Mn-Sr(10) |
Mn-Ti(10) |
|
Mn-V(10) |
Mn-W(10) |
Mn-Y(10) |
Mn-Zn(10) |
Mn-Zr(10) |
Mo-Nb(10) |
Mo-Ni(10) |
|
Mo-O(10) |
Mo-Si(10) |
Mo-Sn(10) |
Mo-Ti(10) |
Mo-V(10) |
Mo-W(10) |
Mo-Y(10) |
|
Mo-Zr(10) |
Na-O(7) |
Na-Si(10) |
Na-Sr(10) |
Na-Zn(10) |
Nb-Ni(10) |
Nb-O(10) |
|
Nb-Si(10) |
Nb-Sn(10) |
Nb-Sr(10) |
Nb-Ti(10) |
Nb-V(10) |
Nb-W(10) |
Nb-Y(10) |
|
Nb-Zr(10) |
Ni-O(10) |
Ni-Pb(10) |
Ni-Sb(10) |
Ni-Si(10) |
Ni-Sn(10) |
Ni-Sr(10) |
|
Ni-Ti(10) |
Ni-V(10) |
Ni-W(10) |
Ni-Y(10) |
Ni-Zn(10) |
Ni-Zr(10) |
O-Pb(10) |
|
O-Sb(7) |
O-Sc(5) |
O-Si(10) |
O-Sn(10) |
O-Sr(5) |
O-Ti(10) |
O-V(10) |
|
O-W(10) |
O-Y(10) |
O-Zn(5) |
O-Zr(10) |
Pb-Sb(10) |
Pb-Si(10) |
Pb-Sn(10) |
|
Pb-Zn(10) |
Pb-Zr(10) |
Sb-Si(10) |
Sb-Sn(10) |
Sb-Zn(10) |
Sc-Si(10) |
Sc-Sn(10) |
|
Sc-Sr(10) |
Sc-Ti(10) |
Sc-W(10) |
Sc-Y(10) |
Sc-Zn(10) |
Sc-Zr(10) |
Si-Sn(10) |
|
Si-Sr(10) |
Si-Ti(10) |
Si-V(10) |
Si-W(10) |
Si-Y(10) |
Si-Zn(10) |
Si-Zr(10) |
|
Sn-Sr(10) |
Sn-Ti(10) |
Sn-V(10) |
Sn-W(10) |
Sn-Y(10) |
Sn-Zn(10) |
Sn-Zr(10) |
|
Sr-Ti(10) |
Sr-V(10) |
Sr-Y(10) |
Sr-Zn(10) |
Ti-V(10) |
Ti-W(10) |
Ti-Zr(10) |
|
V-W(10) |
V-Y(10) |
V-Zn(10) |
V-Zr(10) |
W-Y(10) |
W-Zr(10) |
Y-Zn(10) |
|
Y-Zr(10) |
Zn-Zr(10) |
|
|
|
|
|
|
Ag-Al-Cu(10) |
Ag-Al-Ge(5) |
Ag-Al-Mg(10) |
Ag-Al-Si(10) |
Ag-Al-Zn(5) |
Ag-Cu-Mg(10) |
|
Al-B-Fe(8) |
Al-Ba-Ce(8) |
Al-Ba-Fe(8) |
Al-Ba-La(5) |
Al-Ba-Mn(5) |
Al-Ba-Si(10) |
|
Al-Ba-Y(5) |
Al-Be-Si(5) |
Al-C-Zr(10) |
Al-Ca-Mg(10) |
Al-Ca-Mn(5) |
Al-Ce-Co(5) |
|
Al-Ce-Cu(10) |
Al-Ce-Fe(10) |
Al-Ce-Mg(10) |
Al-Ce-Mn(8) |
Al-Ce-Ni(8) |
Al-Ce-Si(8) |
|
Al-Co-Cu(10) |
Al-Co-Fe(10) |
Al-Co-Mn(5) |
Al-Co-Si(10) |
Al-Cr-Fe(10) |
Al-Cr-Mg(5) |
|
Al-Cr-Mn(5) |
Al-Cr-Ni(10) |
Al-Cr-Si(10) |
Al-Cu-Er(5) |
Al-Cu-Fe(10) |
Al-Cu-Li(10) |
|
Al-Cu-Mg(10) |
Al-Cu-Mn(5) |
Al-Cu-Mo(5) |
Al-Cu-Sb(5) |
Al-Cu-Sc(5) |
Al-Cu-Si(10) |
|
Al-Cu-Zn(10) |
Al-Er-Fe(5) |
Al-Er-Mg(10) |
Al-Er-Mn(5) |
Al-Fe-La(8) |
Al-Fe-Mg(10) |
|
Al-Fe-Mn(10) |
Al-Fe-Mo(5) |
Al-Fe-Ni(10) |
Al-Fe-Si(10) |
Al-Fe-Y(8) |
Al-Fe-Zn(10) |
|
Al-Gd-Mn(8) |
Al-Gd-Ni(5) |
Al-Ge-Si(5) |
Al-La-Mg(5) |
Al-La-Mn(5) |
Al-La-Si(10) |
|
Al-La-Y(5) |
Al-Li-Mg(10) |
Al-Li-Si(10) |
Al-Li-Zn(10) |
Al-Mg-Mn(5) |
Al-Mg-Mo(5) |
|
Al-Mg-Na(5) |
Al-Mg-Sb(5) |
Al-Mg-Sc(5) |
Al-Mg-Si(10) |
Al-Mg-V(10) |
Al-Mg-Y(5) |
|
Al-Mg-Zn(10) |
Al-Mn-Sc(5) |
Al-Mn-Si(10) |
Al-Mn-Y(5) |
Al-Mo-Si(10) |
Al-Si-Sn(5) |
|
Al-Si-Sr(10) |
Al-Si-Ti(10) |
Al-Si-Y(5) |
Al-Si-Zn(10) |
Al-Si-Zr(10) |
Bi-Sn-Zn(10) |
|
Cu-Mg-Si(10) |
Cu-Mg-Zn(10) |
Fe-Mn-Si(5) |
Li-Mg-Si(10) |
Mg-Si-Zn(5) |
|
Database Validation
The PanAl database is validated by a large amount of phase equilibrium data. Two examples are shown here. Figure 1.1 shows the calculated isotherm of Al-Cu-Mg-Si at 500ºC with Si content of 1.8wt%. The experimental data of D.P. Smith [1962Smi] are plotted on it for comparison. Figure 1.2 shows the calculated isopleth of Al-Fe-Mg-Si at 4wt.%Mg and 0.5wt.%Fe with experimental data from Phillips [1961Phi].
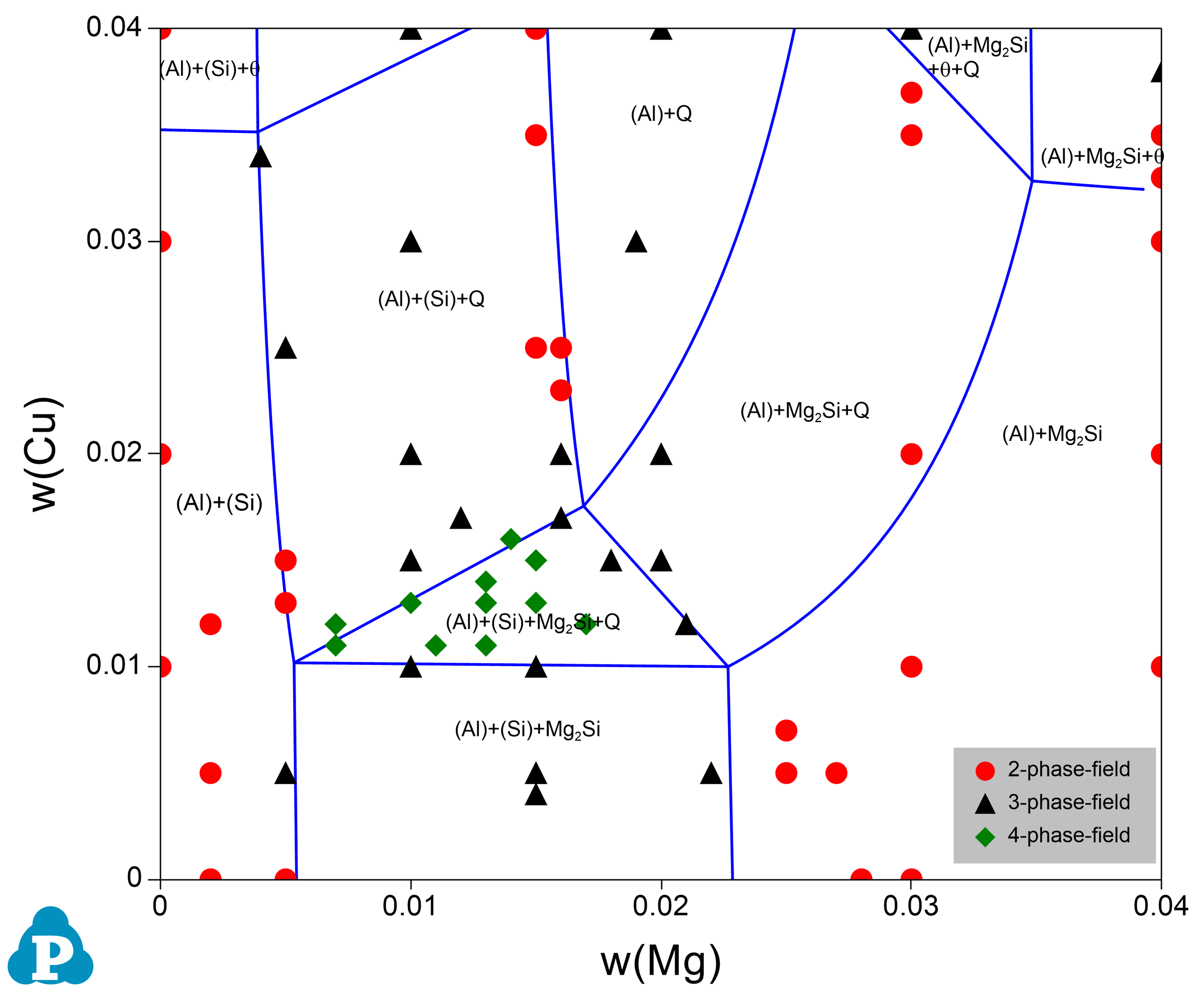
Figure 1.1: Comparison of a calculated isothermal section of Al-Cu-Mg-Si at 1.8wt%Si and at T=500 °C with the experimental data [1962Smi]
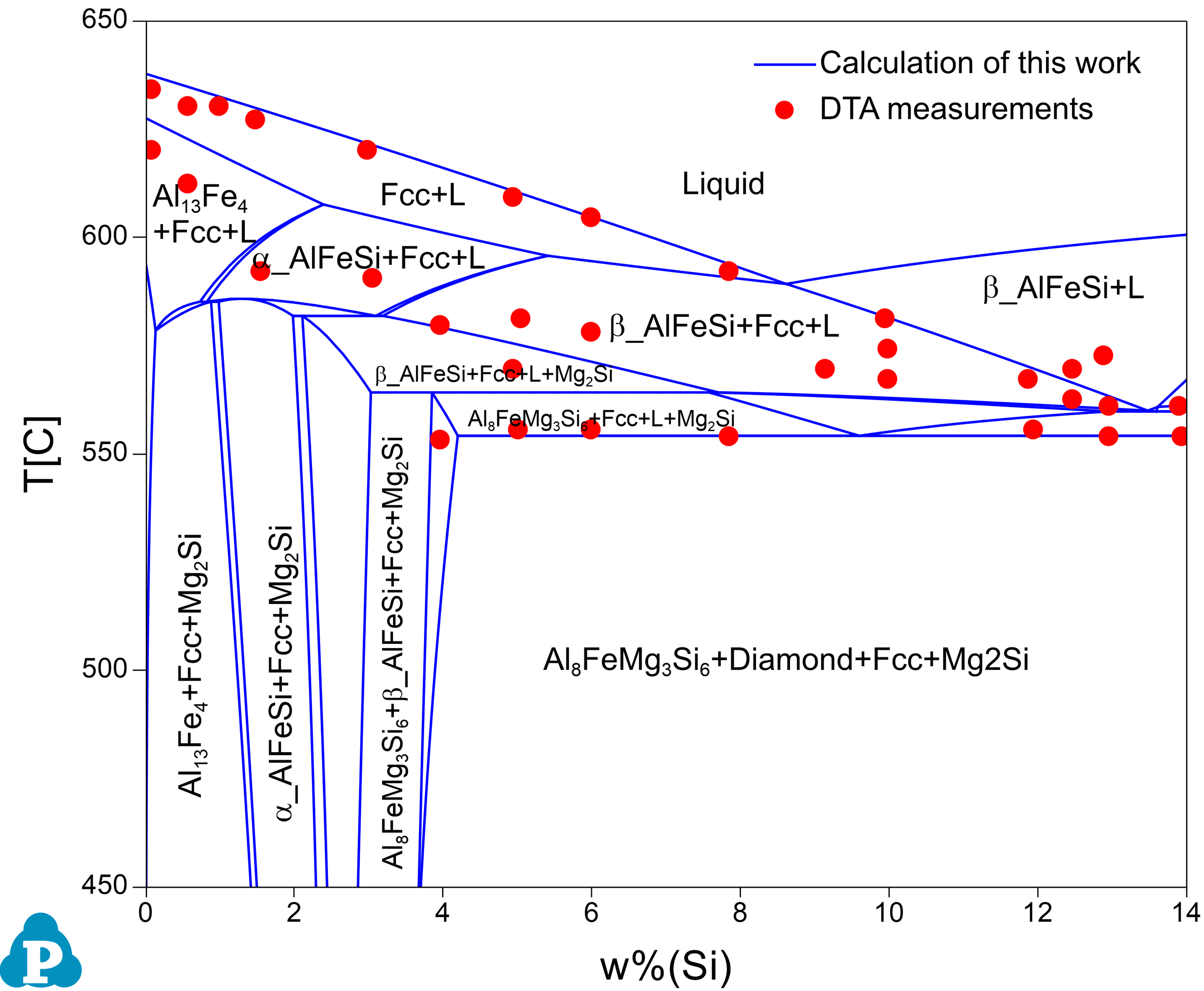
Figure 1.2: Comparison of a calculated isopleth of Al-Fe-Mg-Si at 4wt.%Mg and 0.5wt.%Fe with the experimental data [1961Phi]
In addition to the validation of phase equilibria, the current database has also been subjected to extensive validation by the solidification data of commercial aluminum alloys. The predicted liquidus and solidus temperatures of cast and wrought are shown in. Figure 1.3 ~ Figure 1.6 with experimental data, respectively.
Cast aluminum alloys: 201, 206, 208, 242, 295, 296, 308, 319, 356, 357, 359, 360, 380, 383, 384, 390, 771, 850
Wrought aluminum alloys: 2014, 2017, 2024, 2036, 2124, 2218, 2219, 2319, 3003, 3004, 3105, 4032, 5052, 5056, 5083, 5086, 5154, 5182, 5356, 5454, 5456, 5457, 6005, 6009, 6010, 6061, 6063, 6066, 6070, 6101, 6151, 6201, 6205, 6351, 6463, 7005, 7039, 7049, 7075, 7178, 7475
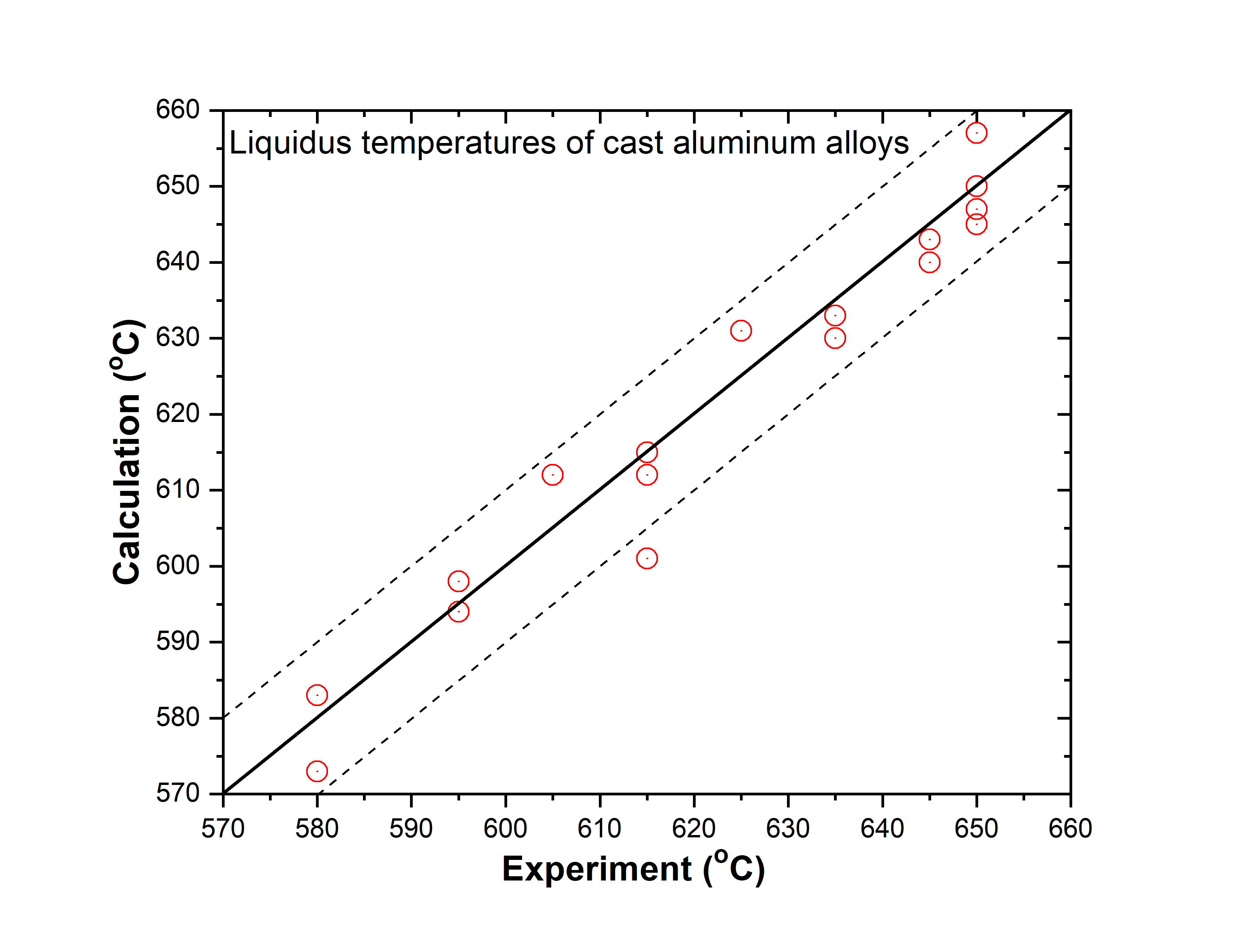
Figure 1.3: Comparison between calculated and experimentally measured liquidus temperatures of cast aluminum alloys
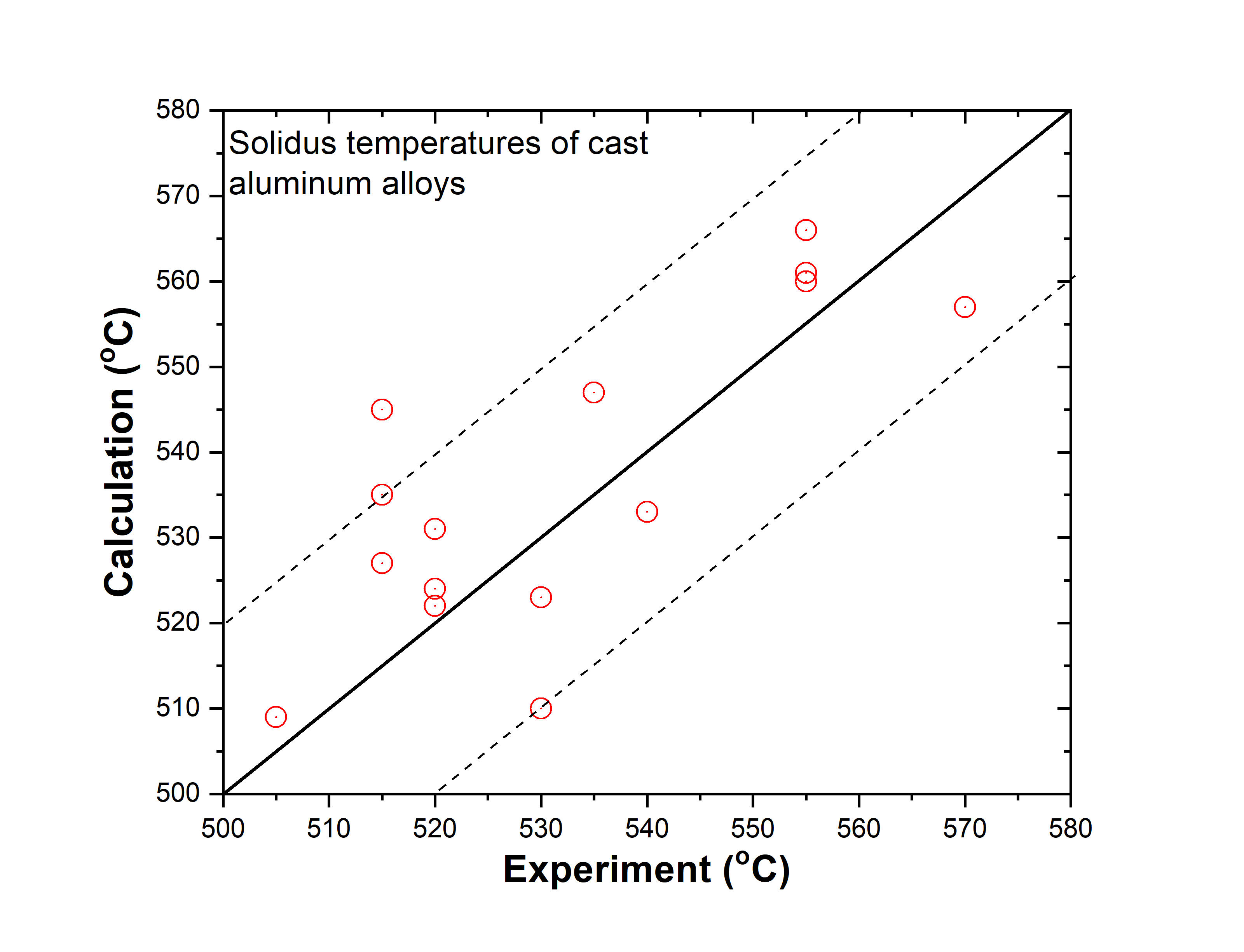
Figure 1.4: Comparison between calculated and experimentally measured solidus temperatures of wrought aluminum alloys
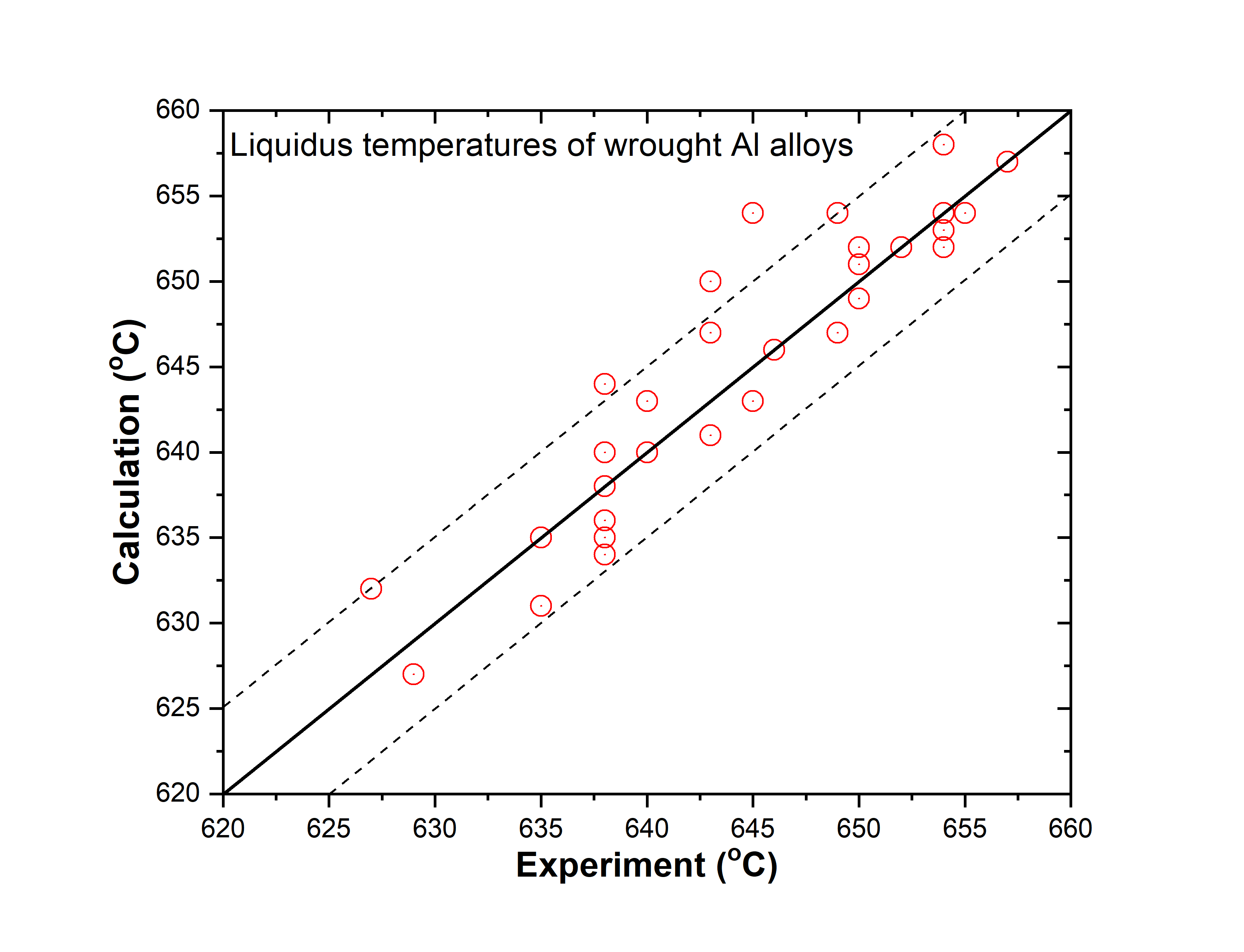
Figure 1.5: Comparison between calculated and experimentally measured liquidus temperatures of wrought aluminum alloys
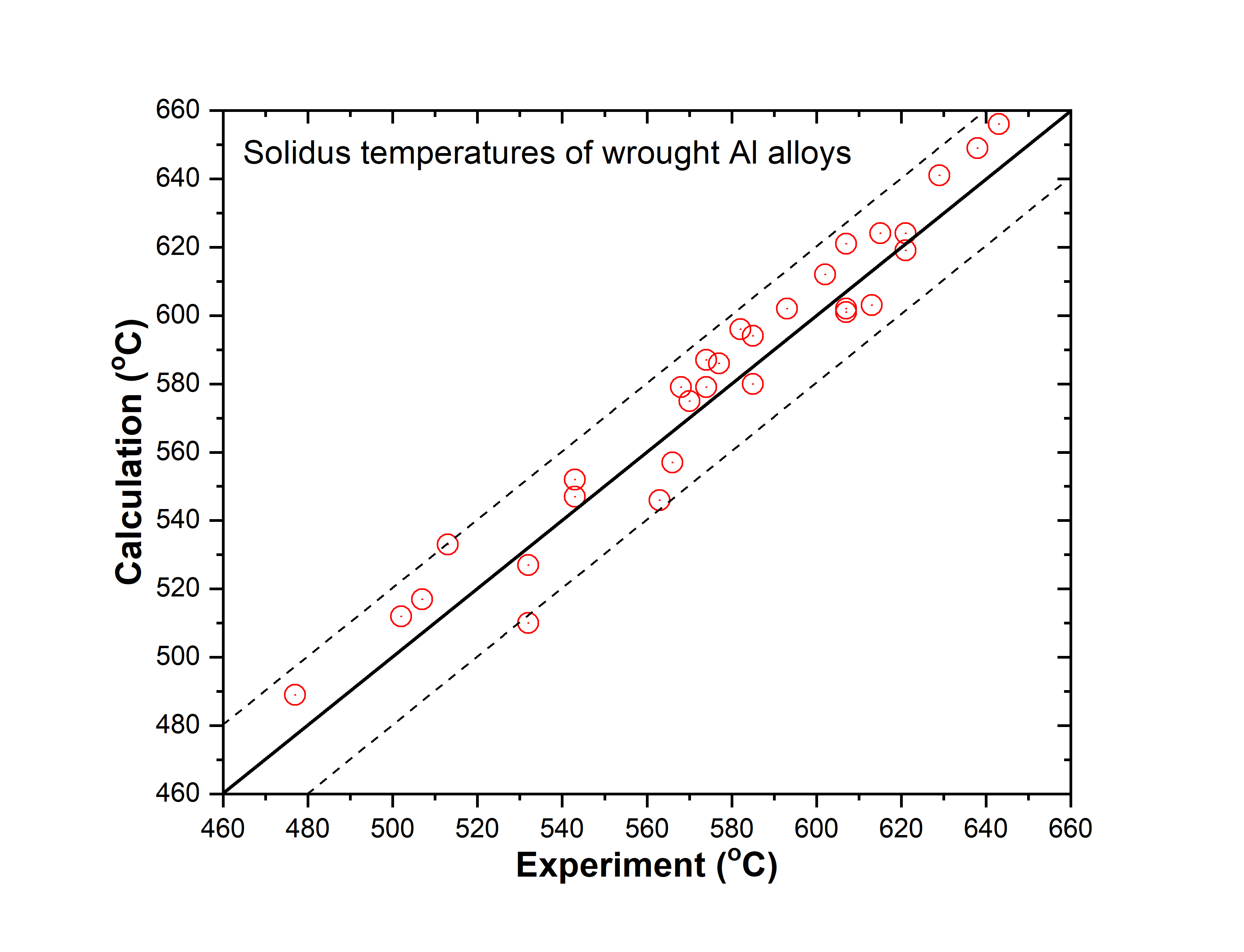
Figure 1.6: Comparison between calculated and experimentally measured solidus temperatures of wrought aluminum alloys
The solidification paths of several commercial aluminum alloys were experimentally investigated by Backerud et al [1986Bac]. The simulated solidification paths using both Scheil model and lever rule for four of these alloys (319.1, A357.2, A6351, A7075) are shown in Figure 1.7 ~ Figure 1.10 with the experimental data of Backerud et al [1986Bac].
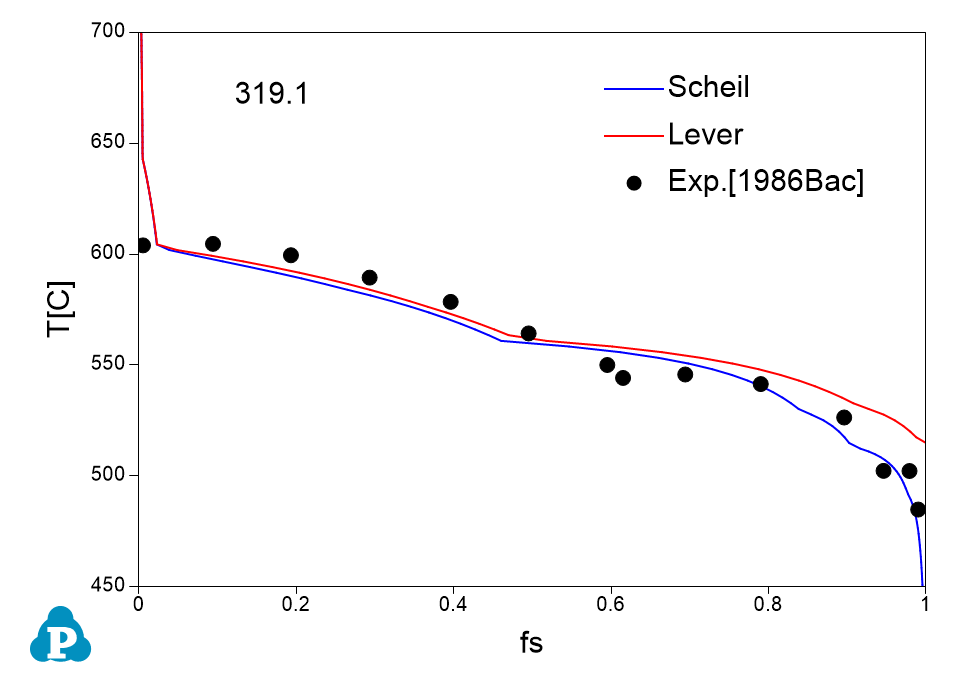
Figure 1.7: Comparison between calculated and experimentally measured solidification paths of 319.1 aluminum alloy
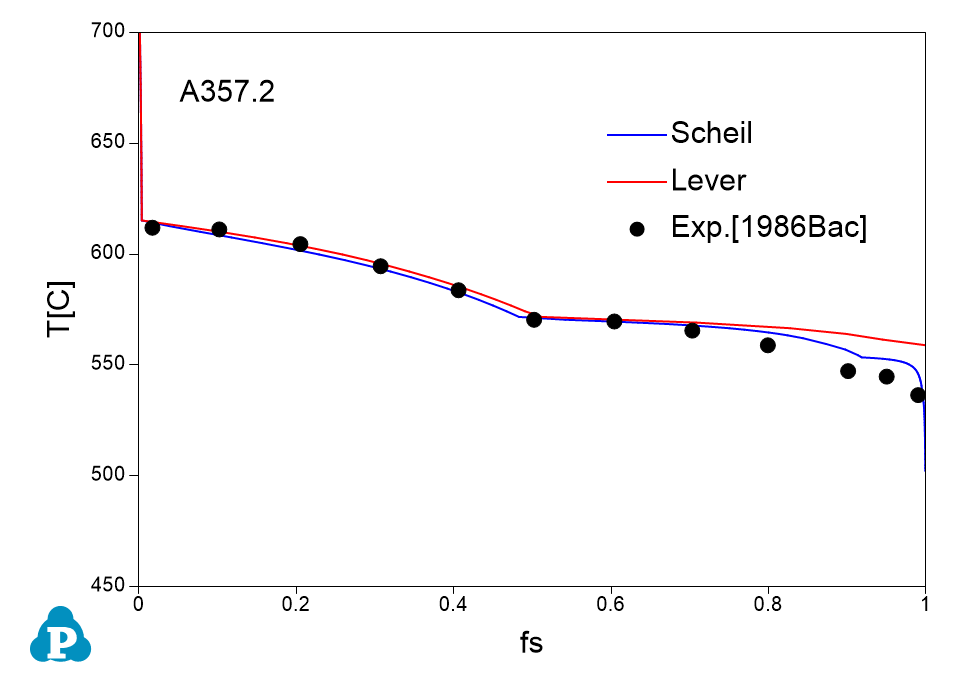
Figure 1.8: Comparison between calculated and experimentally measured solidification paths of A357.2 aluminum alloy
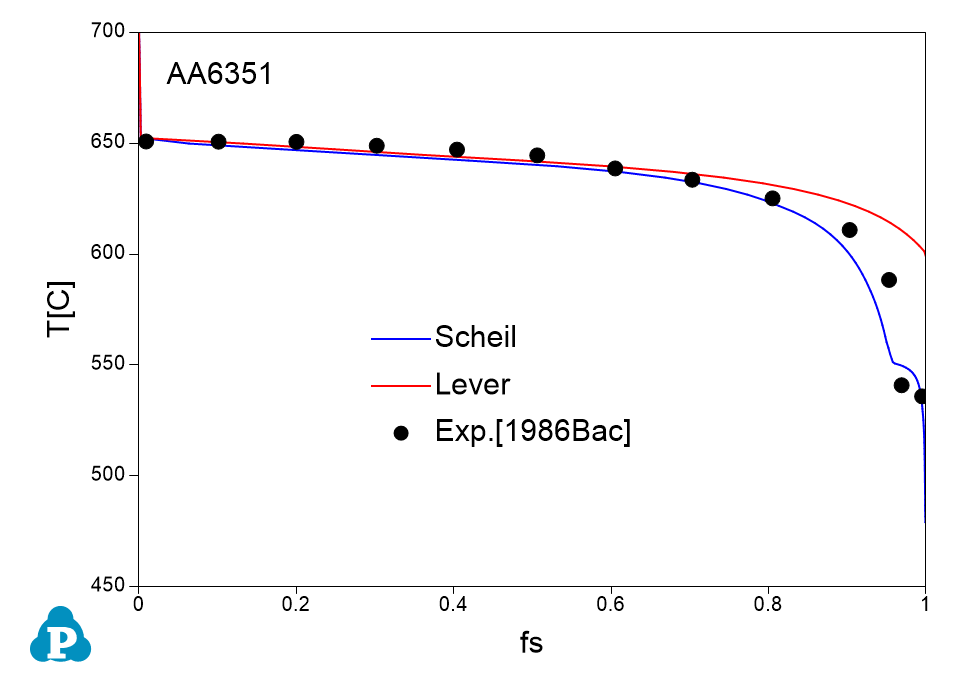
Figure 1.9: Comparison between calculated and experimentally measured solidification paths of AA6351 aluminum alloy
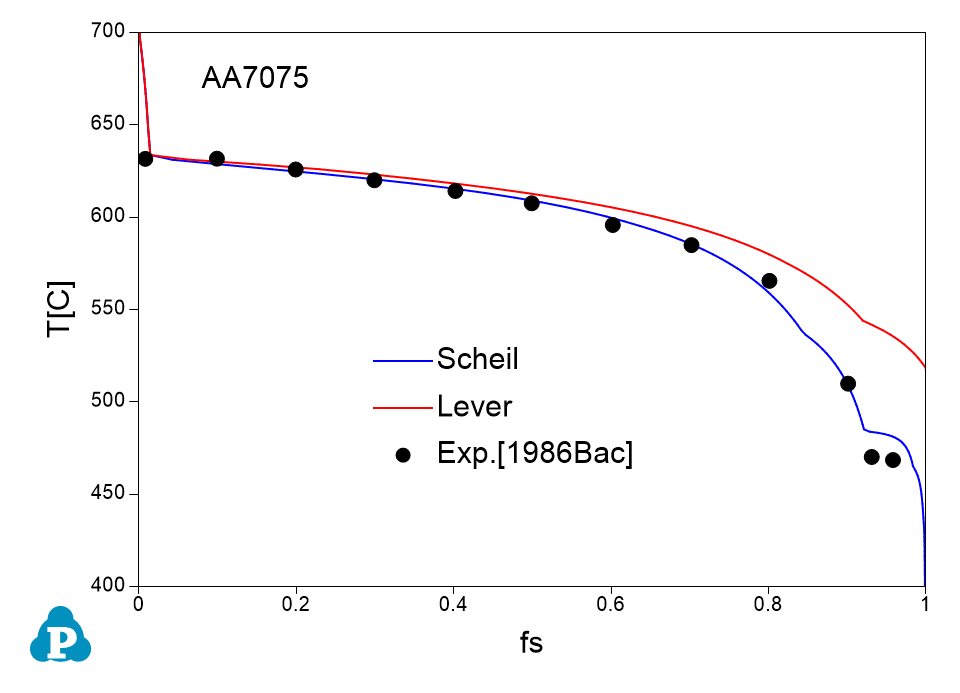
Figure 1.10: Comparison between calculated and experimentally measured solidification paths of AA7075 aluminum alloy
This database can also supply important parameters for processing simulation. One of these parameters is partition coefficient. The calculated partition coefficients have also been extensively validated. Examples for two Al-Cu-Mg-Zn quaternary alloys are given below. Figure 1.11 and Figure 1.12 show comparisons between calculated and measured partition coefficient for Al-4Cu-0.9Mg-2.6Zn (wt%) and Al-2.5Cu-1.3Mg-2.63Zn (wt%), respectively. The good agreement between the experimental and calculated results, as shown in these figures, indicates the reliability of the current PanAl thermodynamic database in providing thermodynamic input for processing simulation.
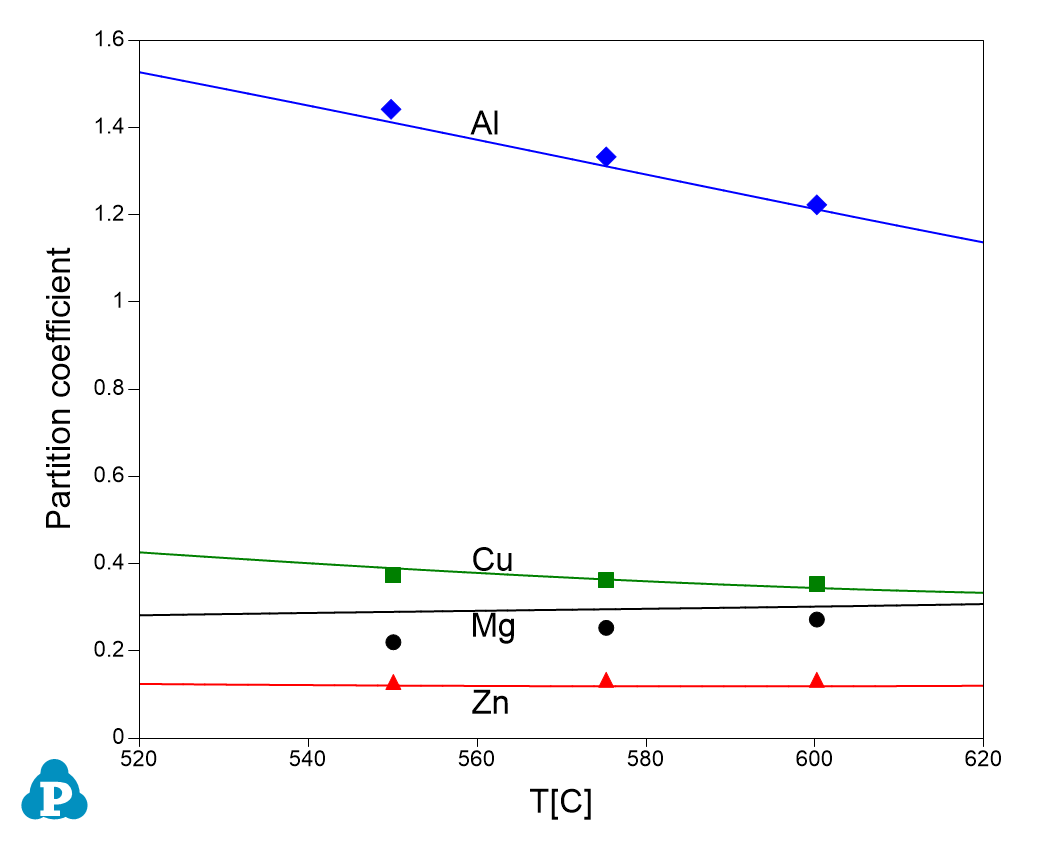
Figure 1.11: Calculated and experimentally determined [1998Lia] partition coefficients of Al-4Cu-0.9Mg-2.6Zn (wt%) alloy
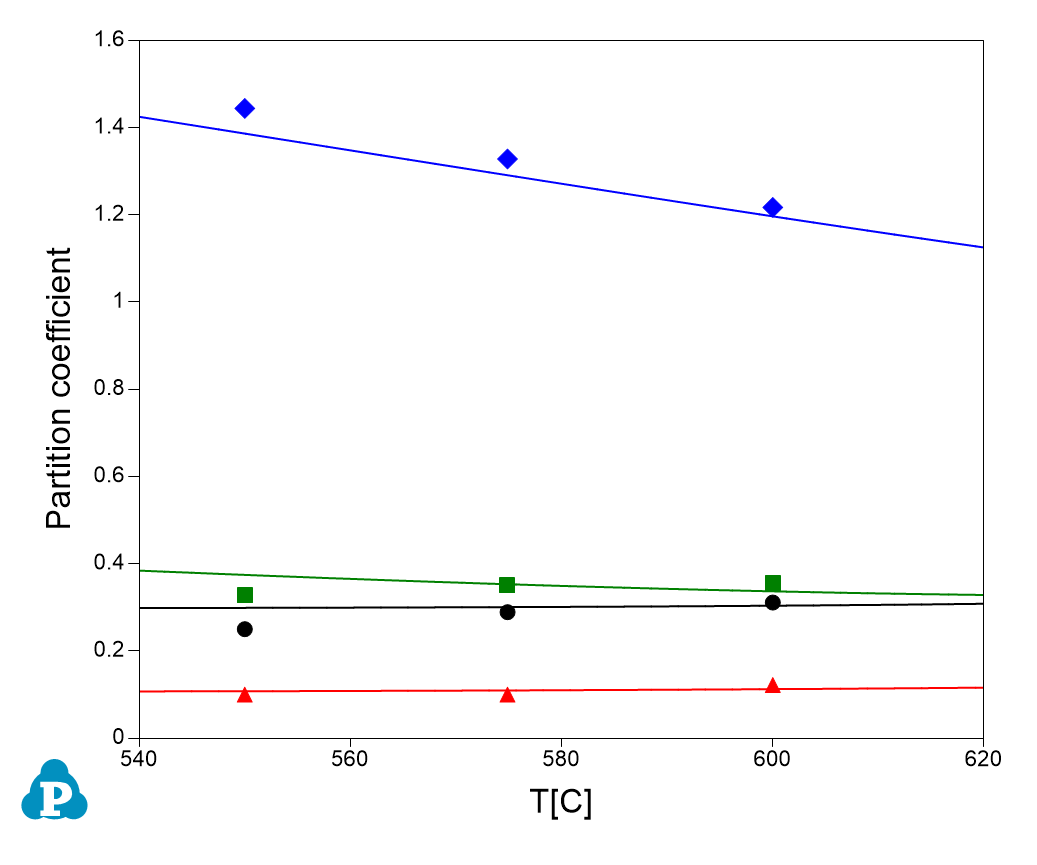
Figure 1.12: Calculated and experimentally determined [1998Lia] partition coefficients of Al-2.5Cu-1.3Mg-2.63Zn (wt%) alloy
[1961Phi] H.W.L. Phillips, Equilibrium diagrams of aluminum alloy systems, The aluminum development association, Information Bulletin 25, London, (1961): 105-108.
[1962Smi] D.P. Smith, Metallurgia, 1(1962): 223-229.
[1986Bac] L.Backerud, et al., Solidification Characteristics of Aluminum Alloys, (1986), Oslo: Tangen Trykk A/S.
[1998Lia] P. Liang, et al., Experimental investigation and thermodynamic calculation of the Al-Mg-Zn system, Thermochim. Acta, 314 (1998): 87-110.
