Example 1.6: Isopleth of Al-Mg-Zn at 15 at% Zn
Purpose: Learn to calculate and use an isoplethal section in a ternary system
Module: PanPhaseDiagram
Thermodynamic Database: Al_Demo.rtdb
Batch file: Example_#1.6.pbfx
As mentioned in Example 1.5: Isotherm of Al-Mg-Zn at 500°C, isopleths are sections vertical to the triangle base plane of the triangular prism. An isopleth is important in understanding phase relationship in a ternary system when temperature is a variable. In this example, we will calculate an isoplethal section in the Al-Mg-Zn ternary parallel to Al-Mg binary with 15 at% of Zn.
Calculation Procedures:
- Load Al_Demo.rtdb following the procedure in Pandat User’s Guide: Load Database, and select Al, Mg and Zn three components;
- Perform 2D calculation following the procedure in Pandat User's Guide: Section Calculation (2D);
- Set Calculation Condition as shown in Figure 1.6.1;
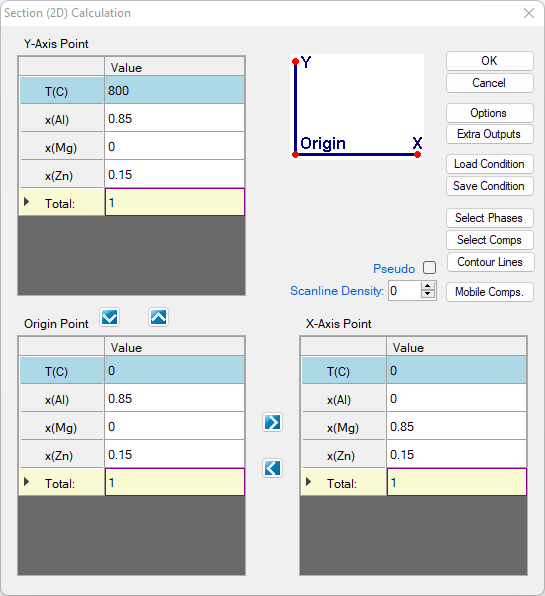
Figure 1.6.1: 2D calculation of an isopleth parallel to Al-Mg binary with 15 at% Zn
Post Calculation Operation:
- Label phase field following the procedure Pandat User’s Guide: Icons for Graph on Toolbar;
- Change graph appearance following the procedure in Pandat User’s Guide: Property;
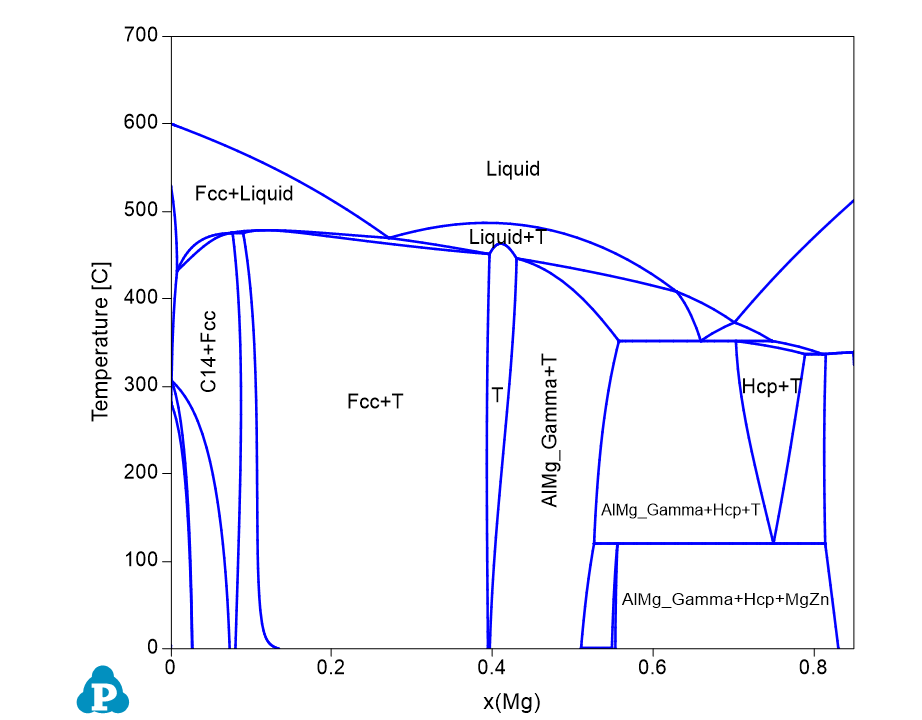
Figure 1.6.2: Calculated isopleth parallel to Al-Mg binary with 15 at% Zn
Information obtained from this calculation:
- Phase stability as a function of composition of Mg and temperature. Single phase, two-phase, and three-phase regions, can be viewed clearly at different composition and temperature (Figure 1.6.2);
- Tie-lines cannot be viewed on an isopleth since they are usually not on the same isoplethal plane except for a pseudo-binary. The tieline information is listed in tieline Table as shown in Figure 1.6.3.
- The invariant reactions and the phase composition for every phase in the invariant reaction are listed in invariant_tieline table as shown in Figure 1.6.4.
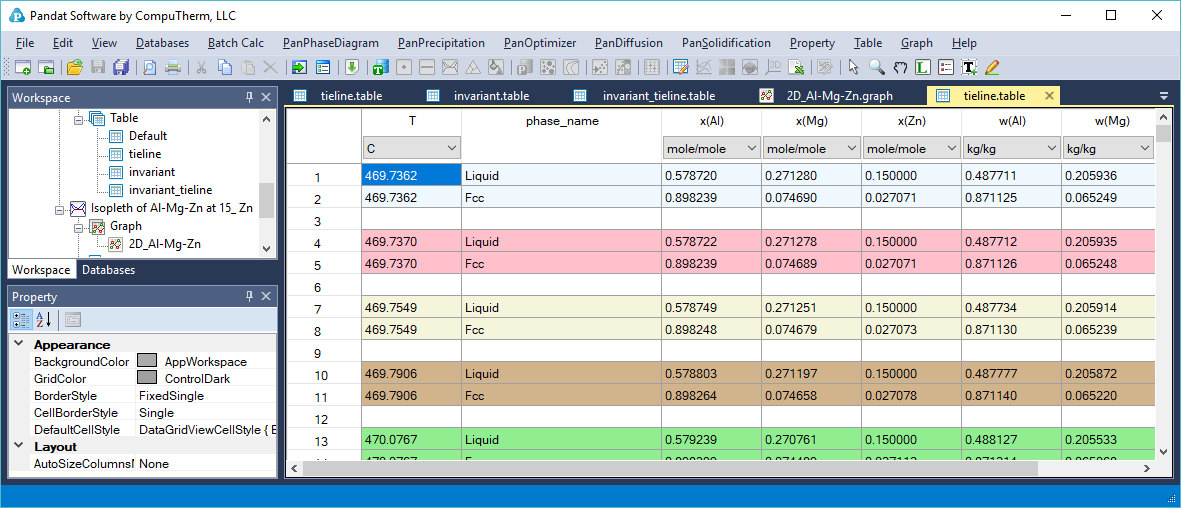
Figure 1.6.3: The tieline table showing the detailed information of calculated tieline
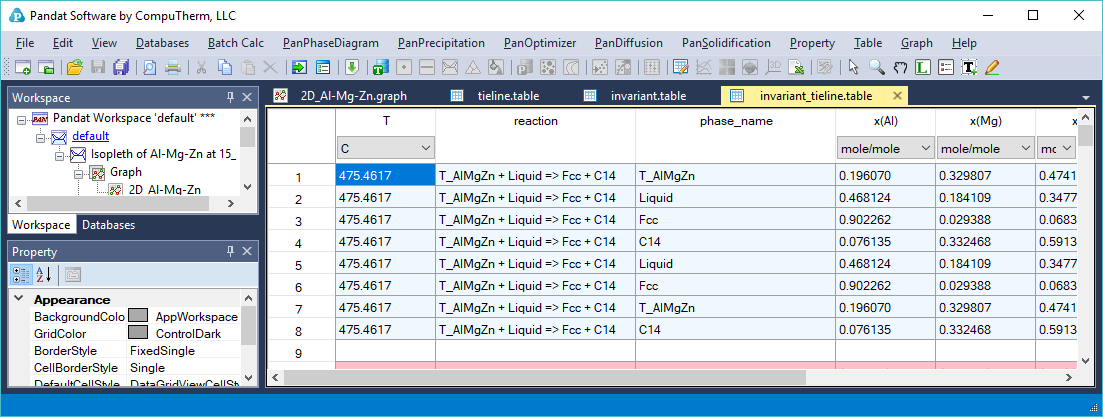
Figure 1.6.4: The invariant_tieline table showing composition of each phase involved in the invariant reaction
