Mobility Database
PanMg2026_MB is an atomic mobility database for Mg-based alloys, which is compatible with the PanMg2026_TH thermodynamic database and suitable for the simulation of diffusion-controlled phenomena using the PanDiffusion module, PanEvolution module, and/or PanSolidification module.
Phases
The atomic mobility within the Liquid, Bcc, Fcc, and Hcp solution phases are assessed in this database.
Self-diffusivity of Pure Elements
The color represents the following meaning:
|
|
: |
Validated |
|
|
: |
Estimated |
|
|
: |
No data |
Table 1: Assessed self-diffusivity of pure elements with different crystal structures
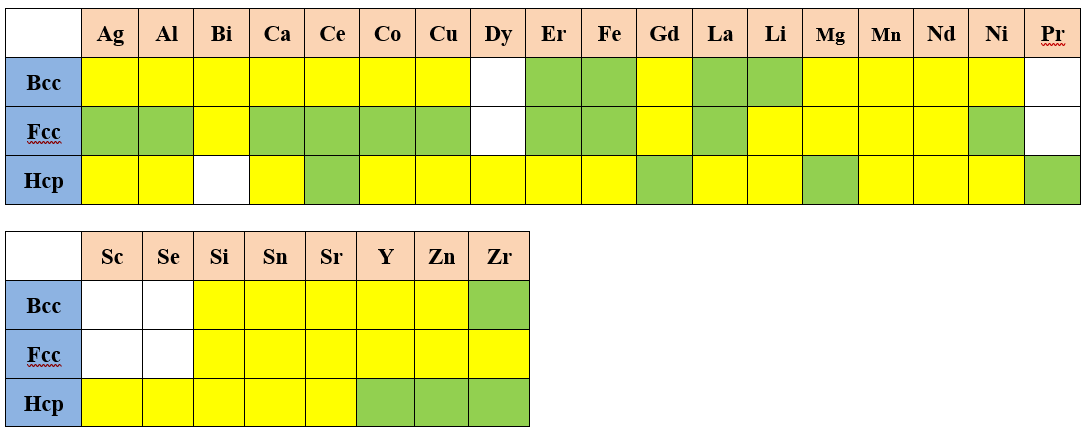
Assessed Systems
In addition to the assessed self-diffusivities shown above, the impurity diffusion data for all included elements in the current PanMg_MB database are also assessed. In the following, the assessed chemical-diffusivity within the binary and ternary systems for the Fcc, and Hcp phases are listed, respectively.
Fcc Phase
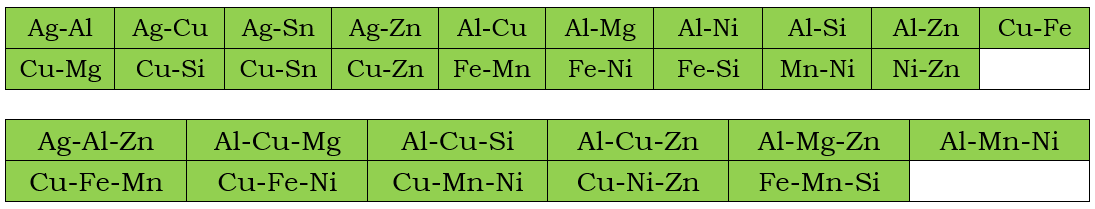
Hcp phase

Database Validation
The simulated concentration profiles of a series of magnesium alloys are used to validate the current mobility database for Mg-based alloys. A few examples of such simulation are shown below.
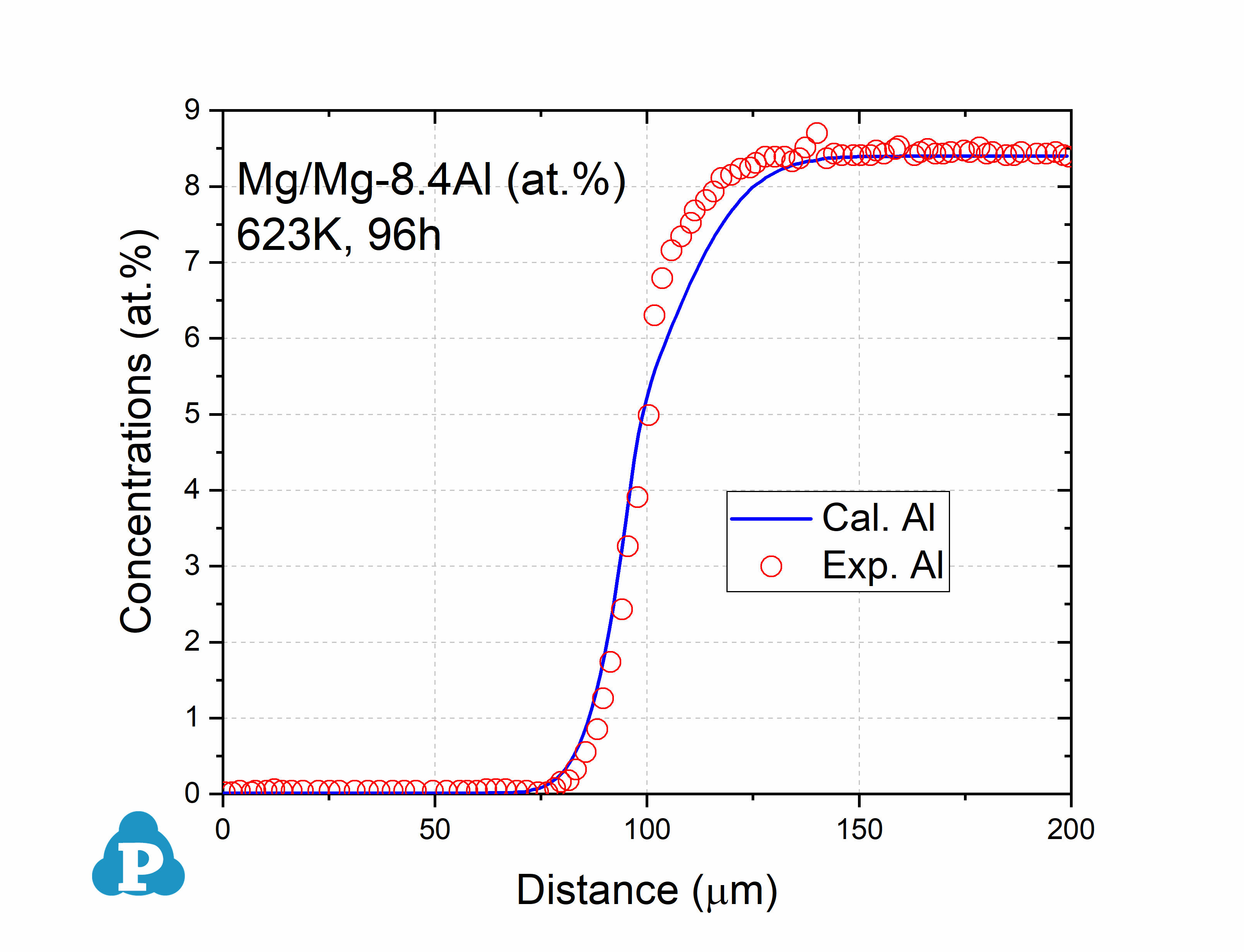
Figure 2.1: Concentration profile of Mg/Mg-8.4Al (at.%) annealed at 623K for 96h with data [2014Kam]
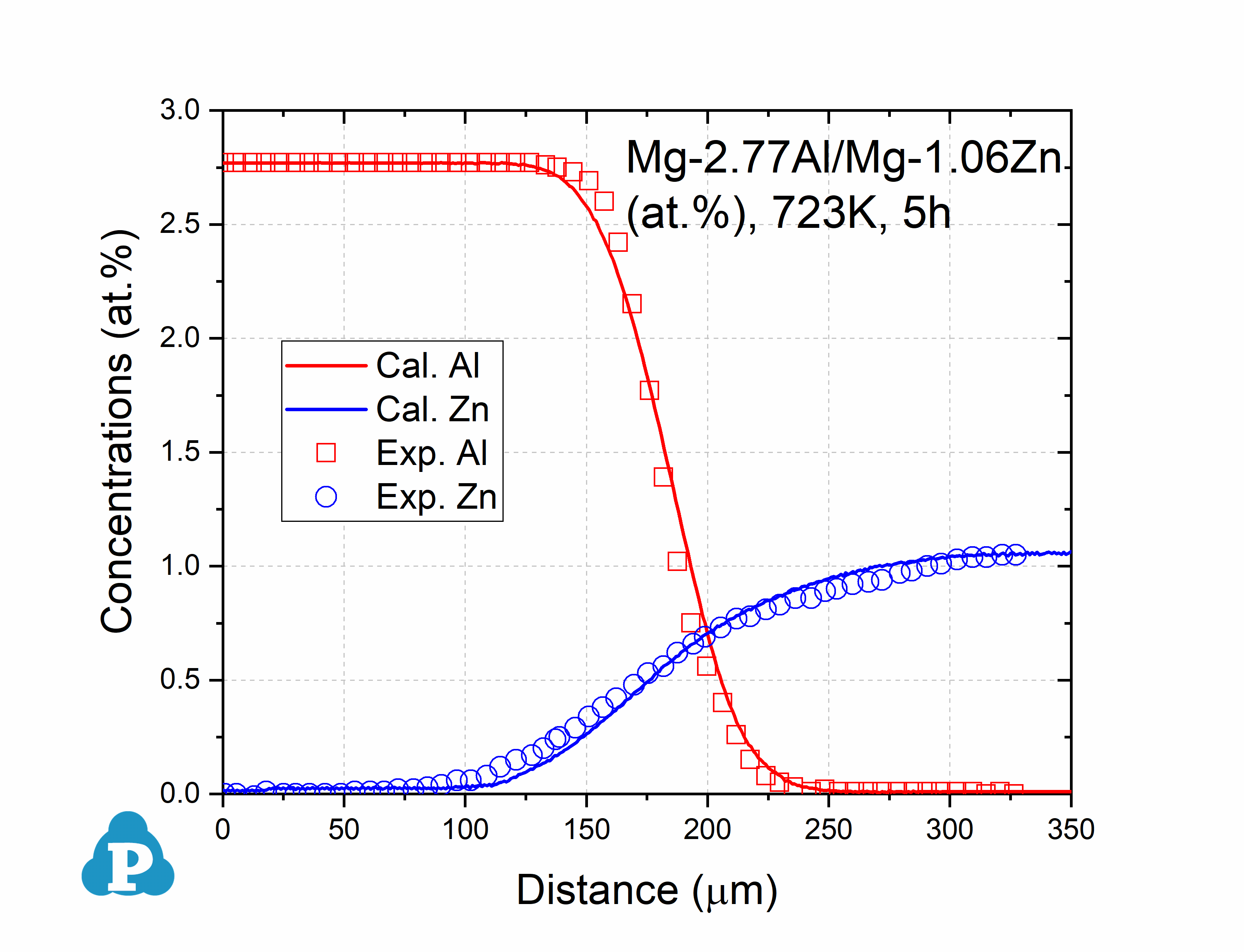
Figure 2.2: Concentration profile of Mg-2.77Al/Mg-1.06Zn (at.%) annealed at 723K for 5h [2016Kam]
Applications
This mobility database is combined with the thermodynamic database for Mg-based alloys, PanMg_TH, to simulate the diffusion-controlled phenomena of Mg-based alloys. A few examples are given below.
Precipitation kinetics of magnesium alloys
The PanEvolution module was developed for the simulation of precipitation kinetics of multi-component alloys. It has been seamlessly integrated with the thermodynamic calculation engine of software, and has been used to simulate the evolution of microstructure and the corresponding mechanical property responses to heat treatment magnesium alloys [2011Cao]. Below shows an example simulation performed for the AZ91 alloy. Figure 2.3 shows the simulated particle size evolution with time of the γ-Mg17Al12 precipitate aged at 200 °C compared with the experimental data of [2001Cel]. Figure 2.4 shows yield strength evolution with time. As is seen, the particles grow and coarsen with ageing time, while the yield strength reaches peak at a time varies with heat treatment temperature. The database used to do this simulation is the combined thermodynamic and mobility database of Mg-based alloys: PanMg_TH+MB. More information regarding to precipitation simulation can be found in module under the Software section.
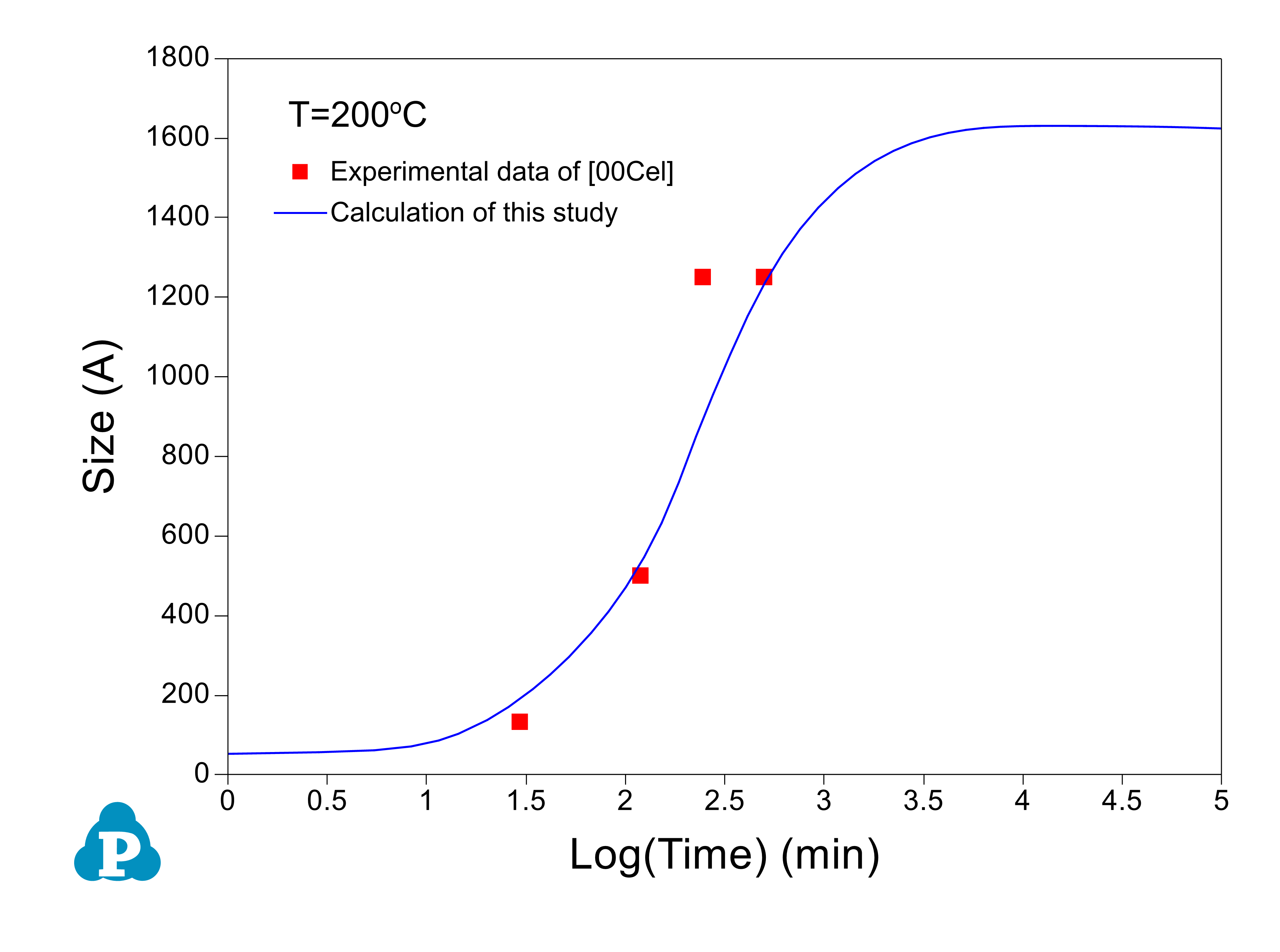
Figure 2.3: Calculated particle size evolution of the γ-Mg17Al12 precipitate aged at 200 °C compared with the experimental data of [2001Cel]
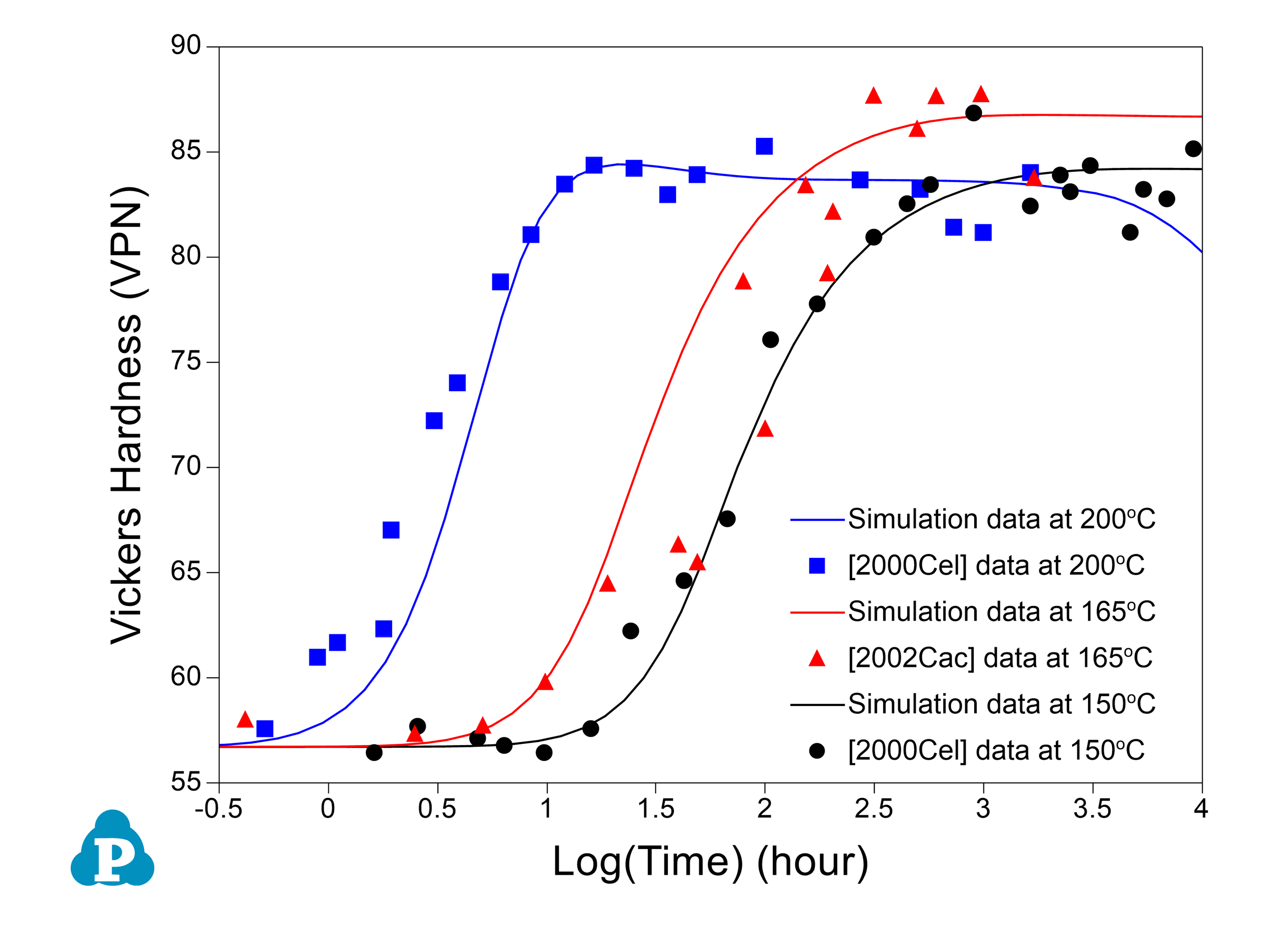
Figure 2.4: Simulated hardness curves of AZ91 alloy aged at 150, 165 and 200 °C compared with the experimentally measured data of [2001Cel, 2002Cac]
Solidification of magnesium alloys
The combined thermodynamic and mobility database of Mg-based alloys: PanMg_TH+MB is also used to simulate the solidification process considering back diffusion in the solid phase [2019Zha]. Figure 2.5 compares the simulated and measured [2013Pal] Al composition profiles in the α(Mg) phase vs. fraction of solid (fs) for Mg-xAl (x=3, 6 ,9) alloys. Figure 2.6 compares the simulated and measured secondary dendrite arm spacing (SDAS) [2013Pal2] results of Mg-Al binary alloys. More information regarding to solidification simulation can be found in module under the Software section.
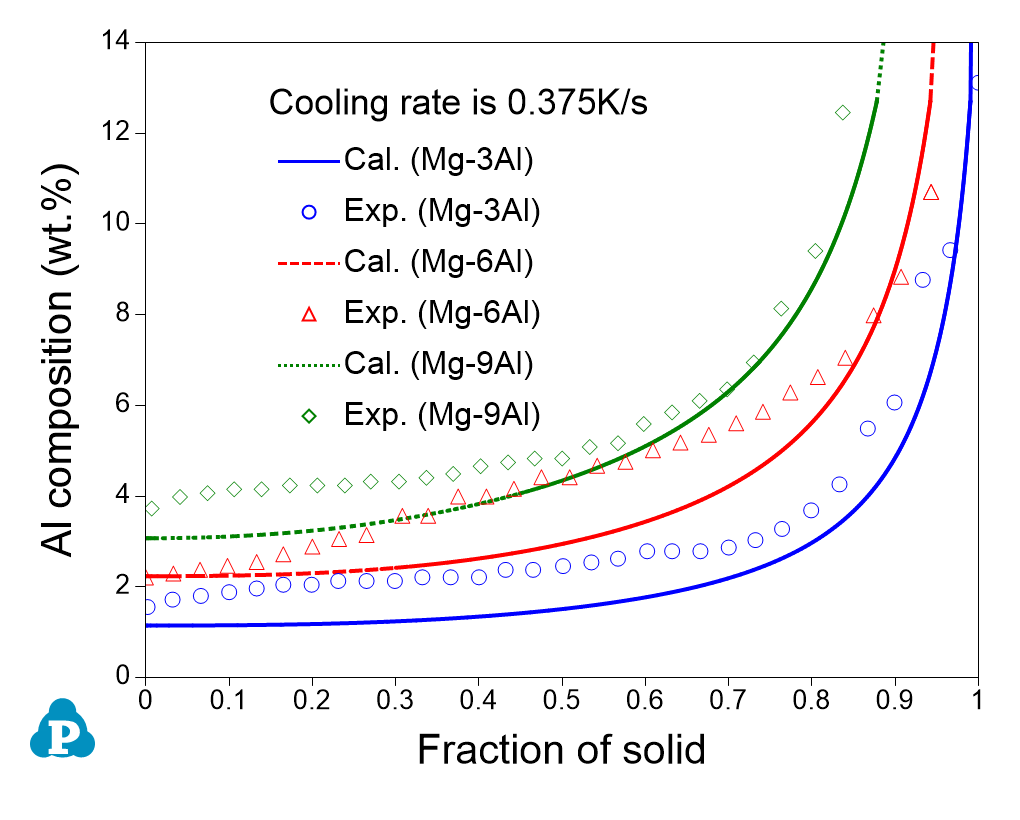
Figure 2.5: Comparison between the simulated and measured [2013Pal] Al composition profiles in the hcp_α(Mg) phase vs. fs for Mg-xAl (x=3, 6 ,9) alloys
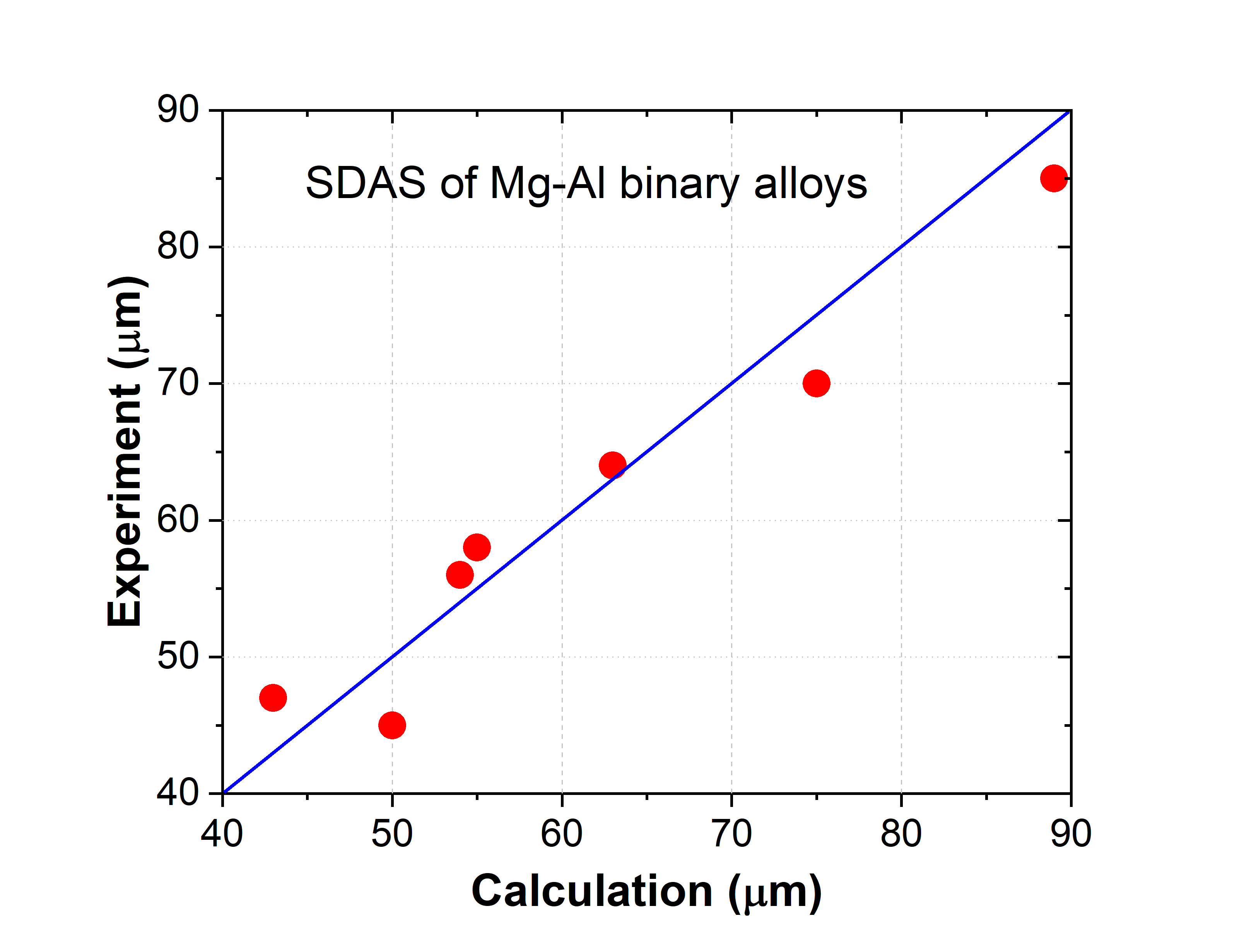
Figure 2.6: Comparison between the simulated and measured SDAS [2013Pal2] for Mg-Al binary alloys
[2001Cel] S. Celotto and T.J. Bastow, Study of precipitation in aged binary Mg–Al and ternary Mg–Al–Zn alloys using 27Al NMR spectroscopy. Acta Materialia, 49(1) (2001): 41-51.
[2002Cac] C.H. Caceres, Hardness and yield strength in cast Mg-Al alloys. AFS Transactions, 110 (2002): 1163-1169.
[2011Cao] W. Cao, et al., An Integrated Computational Tool for Precipitation Simulation. JOM, 63(7) (2011): 29-34
[2013Pal] M. Paliwal, Microstructural development in Mg alloys during solidification: an experimental and modeling study, in Department of Mining and Materials Engineering 2013, McGill University: Montreal, QC.
[2013Pal2] M. Paliwal and I.-H. Jung, The evolution of the growth morphology in Mg–Al alloys depending on the cooling rate during solidification. Acta Materialia, 61(13) (2013): 4848-4860.
[2014Kam] C.C. Kammerer, et al., Interdiffusion and impurity diffusionin polycrystalline Mg solid solution with Al or Zn. Journal of Alloys and Compounds, 617 (2014): 968-974.
[2016Kam] C.C. Kammerer, et al., Interdiffusion in Ternary Magnesium Solid Solutions of Aluminum and Zinc. Journal of Phase Equilibria and Diffusion, 37(1) (2016): 65-74.
[2019Zha] C. Zhang, et al., CALPHAD-Based Modeling and Experimental Validation of Microstructural Evolution and Microsegregation in Magnesium Alloys During Solidification. Journal of Phase Equilibria and Diffusion, 40(4) (2019): 495-507.
