Example 1.4: Point Calculation at Fixed Temperature and Composition
Purpose: Learn to calculate and get detailed phase stability information at a given temperature and composition
Module: PanPhaseDiagram
Thermodynamic Database: Al_Demo.rtdb
Batch file: Example_#1.4.pbfx
From the Al-Mg phase diagram shown in Figure 1.4.1, we know that two phases, AlMg_Beta+Fcc, are stable at the point indicated by Φ (the cross of the two dash lines). Other than that, no detail information about this point can be read directly from this phase diagram. In this example, we perform a Point Calculation to obtain the detail information at the selected point.
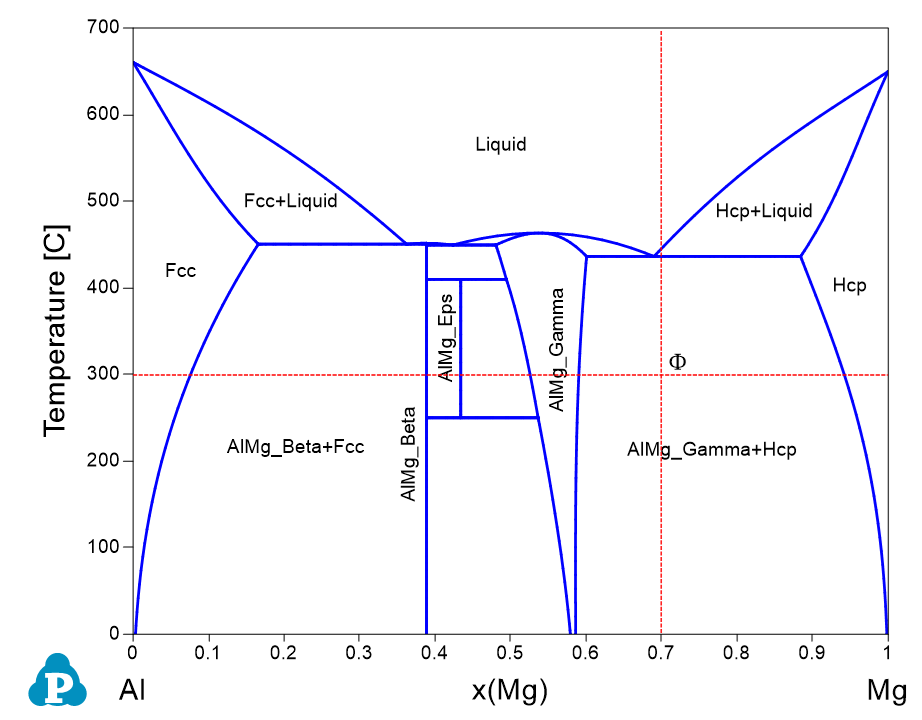
Figure 1.4.1: Al-Mg binary phase diagram
Calculation Procedures:
- Load Al_Demo.rtdb following the procedure in Pandat User’s Guide: Load Database, and select Al and Mg two components;
- Perform 0D calculation following the procedure in Pandat User’s Guide: Point Calculation (0D);
- Set Calculation Condition as shown in Figure 1.4.2;
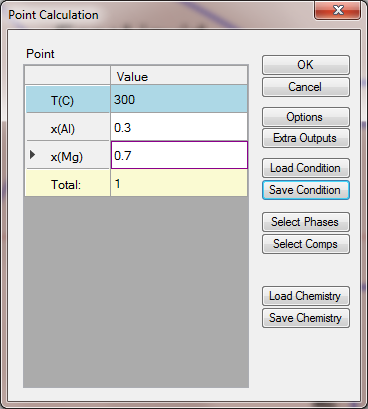
Figure 1.4.2: Point Calculation for an alloy with 30 at% Al and 70 at% of Mg at 300°C
Calculation Method 2 from labeling:
- After the Al-Mg phase diagram is calculated, click
 on the Labeling mode, then put the cursor at the selected point and press Ctrl button, click the mouse.
on the Labeling mode, then put the cursor at the selected point and press Ctrl button, click the mouse.
Information obtained from this calculation:
- Two phases, AlMg_Gamma and Hcp, are stable at the given condition (temperature and composition);
- The G, H, S, and Cp of the system at the given composition and temperature are -22744.9 J/mol, 6539.3 J/mol, 51.0935 J/K·mol, and 30.2876 J/K·mol, respectively;
- The equilibrium composition of AlMg_Gamma is x(Al) = 0.409686, x(Mg) = 0.590314;
- The mole fraction of AlMg_Gamma is 0.687999 (weight fraction is 0.696044);
- The G, H, Cp,
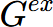 , and
, and 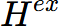 of AlMg_Gamma at equilibrium composition are -23186.3 J/mol, 5976.71 J/mol, 31.3752 J/K·mol, 135.828 J/mol, and 146.6 J/mol, respectively;
of AlMg_Gamma at equilibrium composition are -23186.3 J/mol, 5976.71 J/mol, 31.3752 J/K·mol, 135.828 J/mol, and 146.6 J/mol, respectively; - The site fraction of AlMg_Gamma are:
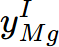 = 1,
= 1, 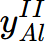 = 0.0291951,
= 0.0291951, 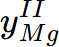 = 0.970805,
= 0.970805, 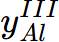 = 0.96088,
= 0.96088, 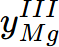 = 0.0391201;
= 0.0391201; - The equilibrium composition of Hcp is x(Al) = 0.058129, x(Mg) = 0.941871;
- The mole fraction of Hcp phase is 0.312001 (weight fraction is 0.303956);
- The G, H, Cp,
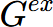 , and
, and 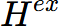 of Hcp at equilibrium composition are -21771.6 J/mol, 7779.87 J/mol, 27.8893 J/K·mol, 179.735 J/mol, and 184.813 J/mol, respectively;
of Hcp at equilibrium composition are -21771.6 J/mol, 7779.87 J/mol, 27.8893 J/K·mol, 179.735 J/mol, and 184.813 J/mol, respectively; - The site fraction of Hcp is the same as its composition since it is essentially one sublattice model, the second sublattice is occupied by vacancy.
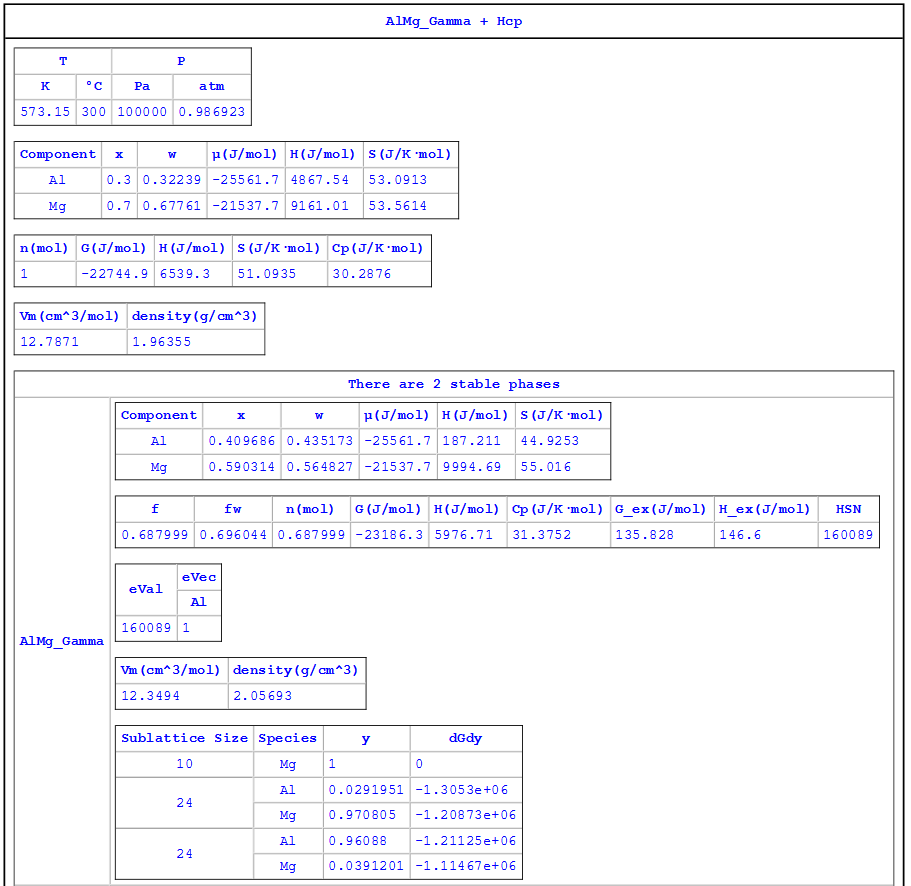
Figure 1.4.3: Calculated results for an alloy with 30 at% Al and 70 at% of Mg at 300°C
