Example 1.3: Phase Fraction as a Function of Temperature
Purpose: Learn to calculate and use a phase fractions vs. temperature plot
Module: PanPhaseDiagram
Thermodynamic Database: Al_Demo.rtdb
Batch file: Example_#1.3.pbfx
In Example 1.2, we have calculated the fraction of each phase as a function of composition. In most cases, people would like to know phase transformation when temperature varies. In this case example, we calculate the fraction of phases as a function of temperature for an AlMg binary alloy with composition of x(Mg)=0.3 (the red dash line in Figure 1.3.1). Such a calculation is especially valuable for a multi-component alloy.
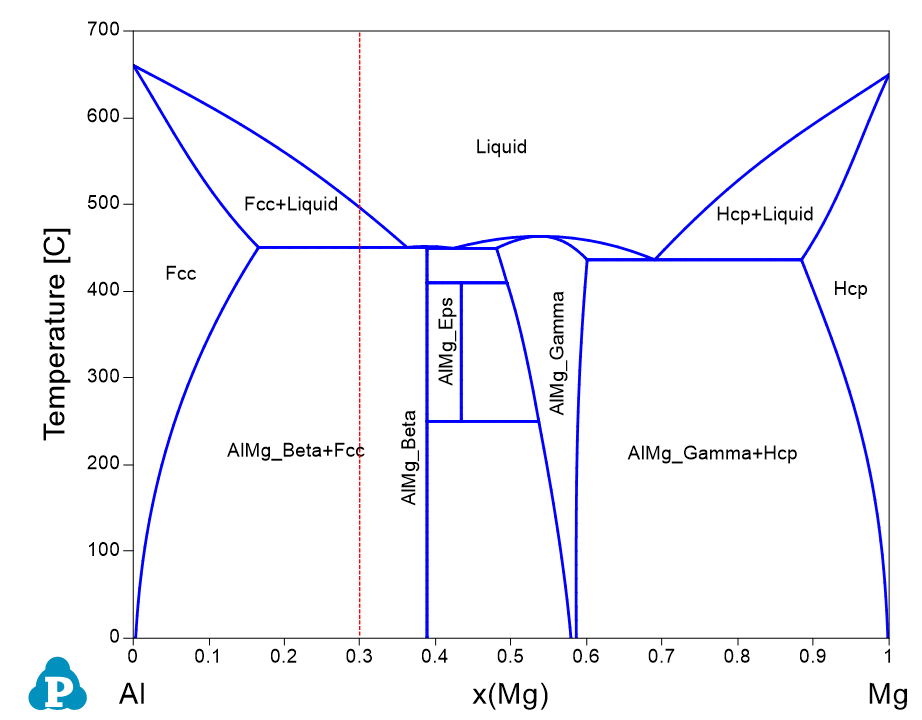
Figure 1.3.1: Al-Mg binary phase diagram
Calculation Procedures:
- Load Al_Demo.rtdb following the procedure in Pandat User’s Guide: Load Database, and select Al and Mg two components;
- Perform 1D calculation following the procedure in Pandat User’s Guide: Line Calculation (1D);
- Set Calculation Condition as shown in Figure 1.3.2;
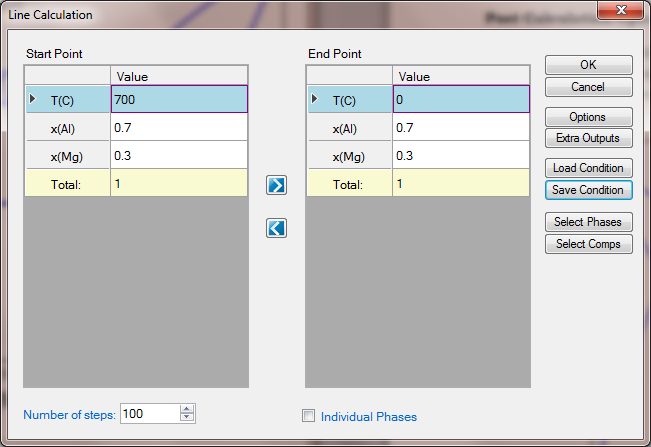
Figure 1.3.2: 1D calculation from 700°C to 0°C for an Al-Mg alloy with x(Mg)=0.3
Post Calculation Operation:
- Add legend for graph following the procedure in Pandat User’s Guide: Icons for Graph on Toolbar;
- Change graph appearance following the procedure in Pandat User’s Guide: Property;
Information obtained from this calculation:
- Liquid is the only stable phase at high temperature until 496.1 °C, which is the liquidus temperature;
- Below liquidus, Fcc phase forms and its fraction increases while that of the Liquid phase decreases until the eutectic temperature 450.47 °C;
- At eutectic temperature 450.47 °C, Liquid is disappeared (drops to 0% from 68.3%), fraction of the AlMg_Beta phase jumps from 0% to 60.21% and that of the Fcc phase jumps from 31.7% to 39.79%;
- Figure 1.3.3 Shows phase fraction vs temperature.
- Details on the fraction of each phase as a function of temperature can also be found in the Default table as shown in Figure 1.3.4;
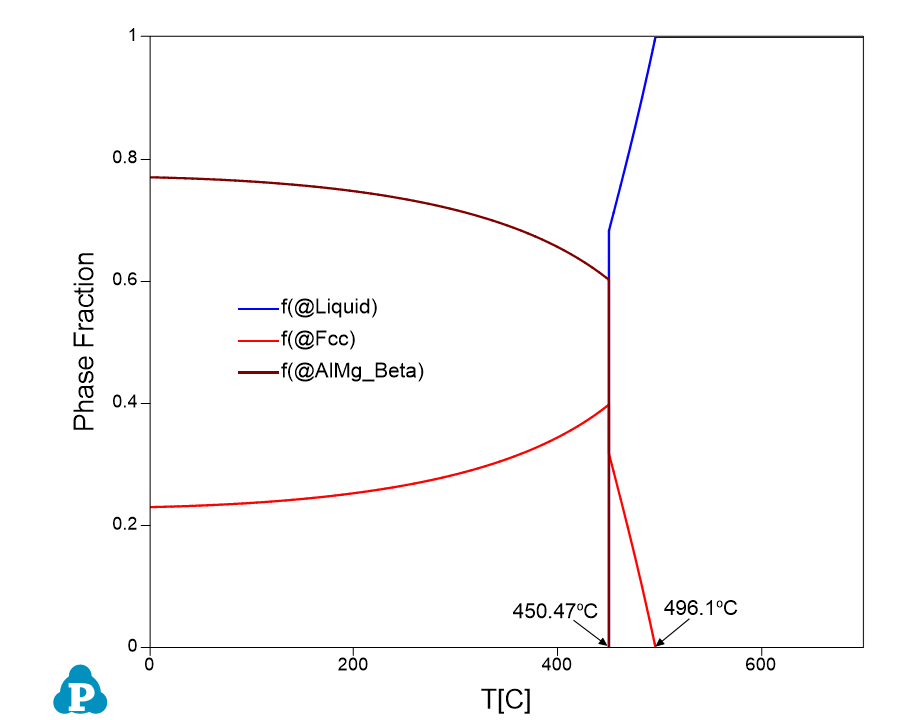
Figure 1.3.3: Phase fraction variation as a function of composition
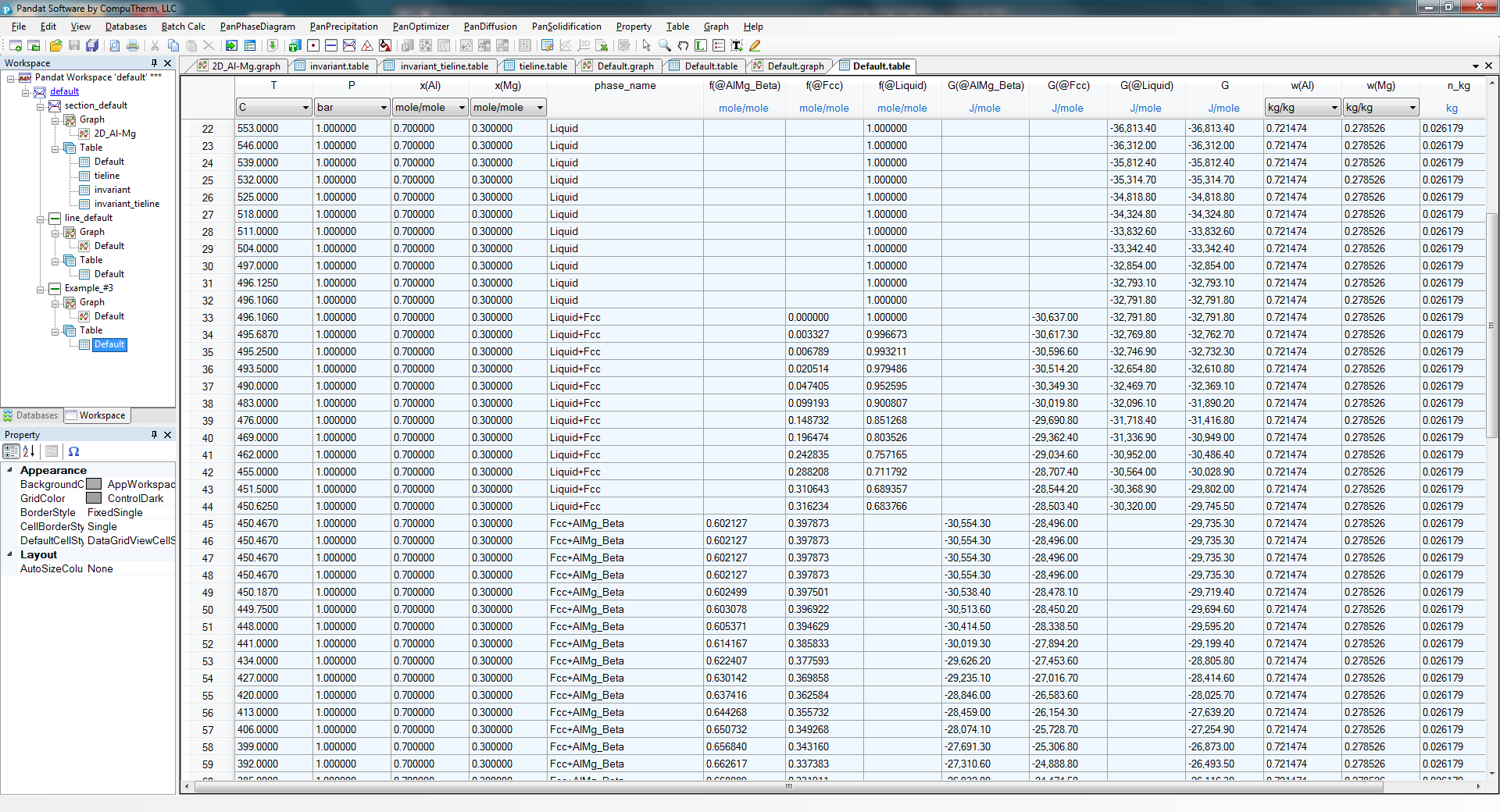
Figure 1.3.4: Default table showing phase fraction variation as a function of temperature
