Tailoring the microstructure and enhancing the corrosion resistance of extruded dilute Mg-0.6Al-0.5Mn-0.25Ca alloy by adding trace Ce
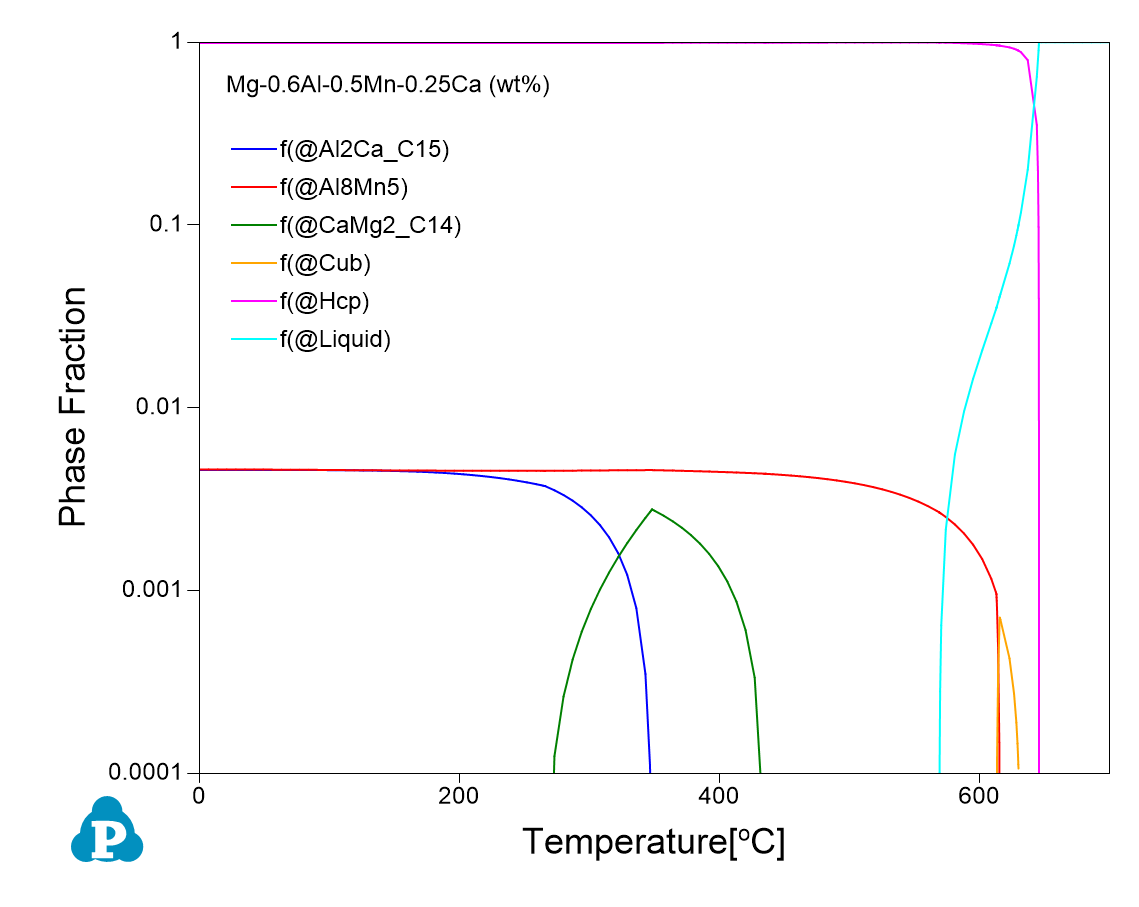
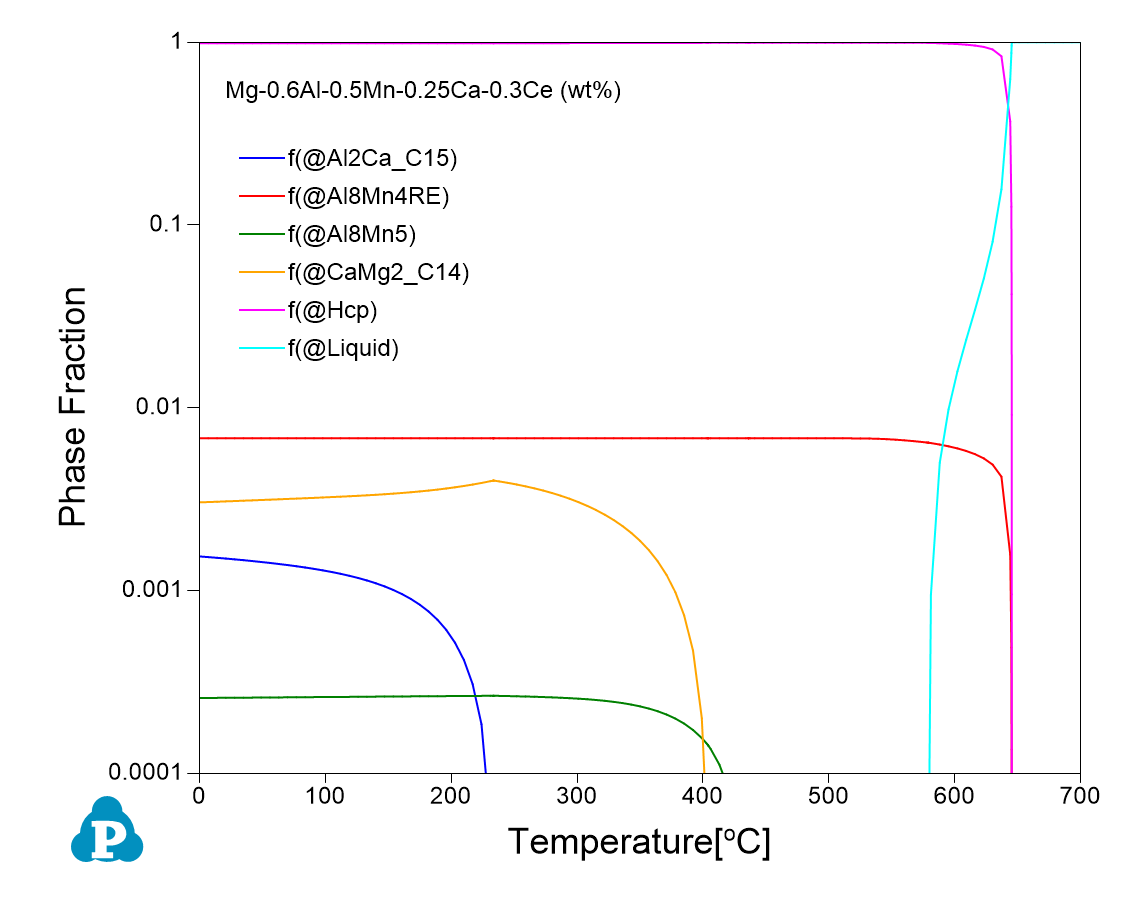
Recent studies have found that addition of rare-earth elements can effectively improve the corrosion resistance of Mg alloys through (1) reducing the harmful effect of impurities (Fe, Cu, and Ni); (2) refining the grains; (3) tailoring the composition, volume fraction, and distribution of the intermetallic phases; and (4) promoting the formation of dense corrosion product films. In a recent publication (Corrosion Science, 2022), Li and colleagues have demonstrated that the corrosion resistance of AMX100 (Mg-0.6Al-0.5Mn-0.25Ca wt%) can be significantly enhanced by adding 0.3 wt% Ce. One of the key reasons that cause corrosion of Mg alloys is the formation of micro galvanic cells between the intermetallic phases and the matrix. Since the Volta potential difference (VPD) between the intermetallic phases and the matrix is the driving force to form the micro galvanic cells, reducing the VPD could help improving the corrosion behavior of Mg alloys. By using Pandat software and PanMg database, the authors found that addition of Ce leads to the formation of the less noble Al8Mn4Ce phase and the reduction of the more noble Al8Mn5 phase. This leads to less driving force to form micro galvanic cells, and therefore improves the corrosion resistance of AMX100.
The following Figure shows equilibrium calculations for two alloy compositions: Mg-0.6Al-0.5Mn-0.25Ca and Mg-0.6Al-0.5Mn-0.25Ca-0.3Ce (all in wt%) using Pandat software and PanMg database. As is seen the addition of 0.3 wt% Ce decreases the amount of the Al8Mn5 phase from ~0.5% to 0.03%.
References:
M.-X. Li, C. Wang, Y.-J. Li, D.-W. Wang, M. Zha, Y. Gao, H.-Y. Wang, Tailoring the microstructure and enhancing the corrosion resistance of extruded dilute Mg-0.6Al-0.5Mn-0.25Ca alloy by adding trace Ce, Corros. Sci., 207 (2022) 110605. doi:10.1016/j.corsci.2022.110605.
Publications related to different topics

